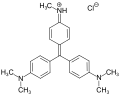
Methyl violet
Methyl violet is a family of organic compounds that are mainly used as dyes. Depending on the number of methyl groups attached to the amine groups, methyl violet can exist in several different forms, such as methyl violet 10B and methyl violet 2B, which are the most common. These dyes are known for their vibrant violet colors and are used in various applications, including as biological stains, pH indicators, and in the dyeing of textiles.
Chemistry[edit]
Methyl violet is a synthetic dye belonging to the family of triarylmethane dyes. The structure of methyl violet dyes consists of two or three methyl groups attached to the nitrogen atoms of the phenyl groups. The chemical formula for the most common form, methyl violet 10B, is C24H28N3Cl. Methyl violet is basic in nature and can form salts with acids, which are often more soluble in water than the free base.
Applications[edit]
Biological Staining[edit]
In microbiology and histology, methyl violet is used as a stain for Gram staining of bacteria, where it is used to differentiate between Gram-positive and Gram-negative bacteria. It is also used in the staining of amyloid plaques in neuroscience.
pH Indicator[edit]
Methyl violet serves as a pH indicator, changing color from yellow at pH 0 to blue-violet at pH 1.6. This property is utilized in various chemical and biological research applications to monitor pH changes.
Textile Dyeing[edit]
Due to its bright color and affinity for fabrics, methyl violet is also used in the textile industry for dyeing purposes. It can be applied to cotton, silk, and paper, among other materials.
Health and Safety[edit]
Exposure to methyl violet dyes can pose health risks, including skin and eye irritation. Ingestion or inhalation of the dye can lead to more severe health issues. Methyl violet has been classified as a potential human carcinogen by some health agencies due to its use in high concentrations and prolonged exposure. Safety measures, such as wearing protective clothing and using proper ventilation, are recommended when handling these dyes.
Environmental Impact[edit]
The use of methyl violet and other synthetic dyes has raised environmental concerns, particularly regarding their persistence and toxicity in aquatic environments. Efforts are being made to develop more environmentally friendly alternatives and to improve the treatment of wastewater containing these dyes.
-
Methyl Violet 2B
-
Methyl Violet 6B
-
Kristallviolett
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian