JWH-051
Overview[edit]
JWH-051 is a synthetic cannabinoid that acts as a potent agonist at the CB1 receptor and CB2 receptor. It is part of the naphthoylindole family of compounds, which are known for their psychoactive properties. JWH-051 was developed in the 1990s by John W. Huffman and his team at Clemson University as part of research into the endocannabinoid system.
Chemical Structure[edit]
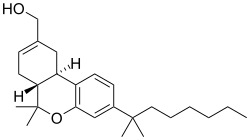
JWH-051 has the chemical formula C25H25NO2 and a molecular weight of 371.47 g/mol. Its structure includes a naphthoyl group attached to an indole core, which is a common feature among synthetic cannabinoids. The presence of a methoxy group on the naphthoyl moiety distinguishes it from other compounds in the JWH series.
Pharmacology[edit]
JWH-051 is a full agonist at both the CB1 and CB2 receptors, with a higher affinity for the CB1 receptor. This receptor is primarily located in the central nervous system, which explains the psychoactive effects of the compound. The CB2 receptor is found mainly in the immune system, and its activation is associated with anti-inflammatory effects.
Mechanism of Action[edit]
The activation of CB1 receptors by JWH-051 leads to the inhibition of adenylate cyclase, resulting in decreased levels of cyclic AMP (cAMP). This cascade of events ultimately affects neurotransmitter release, leading to the compound's psychoactive effects.
Legal Status[edit]
Due to its psychoactive properties, JWH-051 is classified as a controlled substance in many countries. It is often included in legislation targeting synthetic cannabinoids, which are sometimes sold as "spice" or "K2".
Safety and Toxicity[edit]
The safety profile of JWH-051 is not well-documented, as it was primarily developed for research purposes. However, synthetic cannabinoids, in general, have been associated with adverse effects such as tachycardia, hypertension, hallucinations, and psychosis.
Related Compounds[edit]
JWH-051 is part of a larger group of synthetic cannabinoids, including JWH-018, JWH-073, and JWH-200. These compounds share similar structures and pharmacological profiles but differ in their potency and receptor selectivity.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian