Isotryptamine
Chemical compound
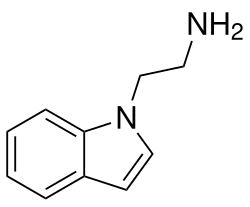
Isotryptamine is a chemical compound that belongs to the class of tryptamines, which are a group of monoamine alkaloids found in various plants, fungi, and animals. Tryptamines are structurally similar to the neurotransmitter serotonin and are known for their role in psychedelic effects when used in certain contexts.
Chemical Structure[edit]
Isotryptamine is a positional isomer of tryptamine, meaning it has the same molecular formula, C10H12N2, but a different arrangement of atoms. The structure of isotryptamine features an indole ring, which is a bicyclic structure consisting of a six-membered benzene ring fused to a five-membered nitrogen-containing pyrrole ring. The side chain of isotryptamine is attached to the indole nitrogen, distinguishing it from other tryptamines where the side chain is typically attached to the carbon adjacent to the nitrogen.
Synthesis[edit]
The synthesis of isotryptamine can be achieved through various chemical methods. One common approach involves the Fischer indole synthesis, which is a chemical reaction that transforms phenylhydrazines and aldehydes or ketones into indoles. This method can be adapted to produce isotryptamine by selecting appropriate starting materials and reaction conditions.
Pharmacology[edit]
The pharmacological properties of isotryptamine are not as well-studied as those of other tryptamines like DMT or psilocybin. However, due to its structural similarity to serotonin, it is hypothesized that isotryptamine may interact with serotonin receptors in the brain. This interaction could potentially lead to effects on mood, perception, and cognition, although specific studies on isotryptamine's effects are limited.
Potential Applications[edit]
Research into isotryptamine and related compounds is ongoing, with interest in their potential applications in psychiatry and neuroscience. Tryptamines have been explored for their therapeutic potential in treating conditions such as depression, anxiety, and post-traumatic stress disorder (PTSD). However, isotryptamine itself has not been the focus of extensive clinical research.
Safety and Legal Status[edit]
The safety profile of isotryptamine is not well-documented, and it is not widely used or available. As with many tryptamines, there may be legal restrictions on its use and distribution, depending on the jurisdiction. It is important for researchers and users to be aware of local laws and regulations regarding tryptamines.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian