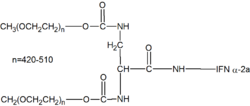Peginterferon alfa-2a
Peginterferon alfa-2a is a pegylated form of interferon alfa-2a, a type of interferon used as an antiviral and antineoplastic agent. It is primarily used in the treatment of hepatitis C and hepatitis B infections. Peginterferon alfa-2a is marketed under the brand name Pegasys.
Mechanism of Action[edit]
Peginterferon alfa-2a works by binding to specific receptors on the surface of cells, initiating a complex cascade of intracellular events. This leads to the activation of multiple genes that inhibit viral replication and modulate the immune response. The pegylation process, which involves attaching a polyethylene glycol (PEG) molecule to the interferon, increases the half-life of the drug, allowing for less frequent dosing compared to non-pegylated interferons.
Indications[edit]
Peginterferon alfa-2a is indicated for the treatment of:
- Chronic hepatitis C (CHC) in patients with compensated liver disease.
- Chronic hepatitis B (CHB) in patients with compensated liver disease and evidence of viral replication and liver inflammation.
Administration and Dosage[edit]
Peginterferon alfa-2a is administered via subcutaneous injection. The dosage and duration of treatment vary depending on the condition being treated and the patient's response to therapy. For chronic hepatitis C, it is often used in combination with ribavirin.
Side Effects[edit]
Common side effects of peginterferon alfa-2a include:
- Flu-like symptoms (fever, chills, muscle aches)
- Fatigue
- Depression
- Insomnia
- Anorexia
- Nausea
- Injection site reactions
Serious side effects can include:
Contraindications[edit]
Peginterferon alfa-2a is contraindicated in patients with:
- Hypersensitivity to interferon alfa or any component of the formulation
- Autoimmune hepatitis
- Decompensated liver disease
Related Pages[edit]
See Also[edit]
References[edit]
External Links[edit]
| Peginterferon alfa-2a | |
|---|---|
|
| |
| Routes of administration | Subcutaneous injection
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian