Hexyl nitrite
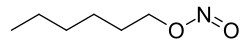
0Hexyl nitrite is a chemical compound that belongs to the class of organic compounds known as alkyl nitrites. It is a colorless liquid with a characteristic fruity odor and is used primarily as a component in certain industrial applications.
Chemical Structure[edit]
Hexyl nitrite has the chemical formula C6H13NO2. It consists of a hexyl group, which is a six-carbon alkyl group, attached to a nitrite functional group. The nitrite group consists of one nitrogen atom and two oxygen atoms, and is responsible for many of the chemical properties of hexyl nitrite.
Properties[edit]
Hexyl nitrite is a volatile liquid at room temperature, with a boiling point of approximately 104 degrees Celsius. It is highly flammable and should be handled with care. It is also a strong oxidizing agent, which means it can cause other substances to lose electrons in a chemical reaction.
Uses[edit]
Hexyl nitrite is used in a variety of industrial applications. It is often used as a component in the manufacture of certain types of rubber and plastic. It is also used as a solvent in some industrial processes.
Health Effects[edit]
Exposure to hexyl nitrite can have a number of health effects. Inhalation can cause dizziness, headache, and unconsciousness. Ingestion can cause nausea, vomiting, and abdominal pain. Long-term exposure can lead to more serious health problems, including damage to the liver and kidneys.
Safety[edit]
Due to its flammability and potential health effects, hexyl nitrite should be handled with care. It should be stored in a cool, well-ventilated area away from heat sources and open flames. Protective clothing, including gloves and eye protection, should be worn when handling hexyl nitrite.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
