Fasoracetam
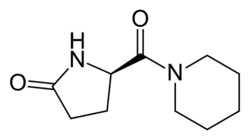
Fasoracetam is a nootropic drug of the racetam family. It is a research chemical that was developed by the Japanese pharmaceutical company Nippon Shinyaku in the 1990s as a potential treatment for Alzheimer's disease.
History[edit]
Fasoracetam was first synthesized in the early 1990s by Nippon Shinyaku, a Japanese pharmaceutical company. The company initially developed the drug as a potential treatment for vascular dementia, but later shifted its focus to Alzheimer's disease. However, clinical trials were discontinued in 1995 due to lack of efficacy.
Pharmacology[edit]
Fasoracetam is a racetam compound, which means it shares a pyrrolidone nucleus with other drugs in this family, such as piracetam and aniracetam. It is believed to work by modulating glutamate, GABA, and acetylcholine neurotransmission. Specifically, fasoracetam has been shown to up-regulate GABA-B receptors and enhance cholinergic function.
Clinical Research[edit]
In recent years, fasoracetam has been the subject of renewed interest in the scientific community. A number of preclinical and clinical studies have been conducted to investigate its potential therapeutic effects. Some research suggests that fasoracetam may have nootropic effects, such as enhancing memory and cognition. Other studies have explored its potential as a treatment for conditions like ADHD and anxiety disorders.
Side Effects[edit]
As with any drug, fasoracetam can cause side effects. These may include headache, fatigue, nausea, and gastrointestinal discomfort. However, these side effects are generally mild and temporary. More research is needed to fully understand the safety profile of fasoracetam.
Legal Status[edit]
Fasoracetam is currently unregulated in many countries, including the United States. This means it can be purchased and used without a prescription. However, it is not approved by the Food and Drug Administration (FDA) for any medical use.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
