Dodecyl gallate
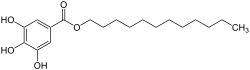

Dodecyl gallate is an organic compound that is classified as an ester of gallic acid and dodecanol. It is commonly used as an antioxidant in the food industry and in cosmetics to prevent rancidity and oxidation.
Chemical Structure and Properties[edit]
Dodecyl gallate has the chemical formula C19H30O5. It is a white to slightly yellowish powder that is soluble in oils and organic solvents but has limited solubility in water. The compound consists of a gallic acid moiety esterified with a dodecyl (lauryl) alcohol chain.
Synthesis[edit]
Dodecyl gallate is synthesized through the esterification of gallic acid with dodecanol. This reaction typically involves the use of an acid catalyst to facilitate the formation of the ester bond.
Applications[edit]
Food Industry[edit]
In the food industry, dodecyl gallate is used as an antioxidant to extend the shelf life of products by preventing the oxidation of fats and oils. It is often added to edible oils, margarine, and butter.
Cosmetics[edit]
Dodecyl gallate is also used in cosmetics and personal care products for its antioxidant properties. It helps to stabilize formulations and prevent the degradation of active ingredients.
Safety and Regulation[edit]
Dodecyl gallate is generally recognized as safe (GRAS) by regulatory agencies when used in accordance with good manufacturing practices. However, it is important to adhere to specified usage limits to avoid potential adverse effects.
Related Compounds[edit]
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
