Demecarium bromide
A medication used in the treatment of glaucoma and other eye conditions
Demecarium bromide[edit]
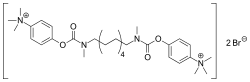
Demecarium bromide is a cholinesterase inhibitor used primarily in the management of glaucoma and other ocular conditions. It is a quaternary ammonium compound that acts by inhibiting the enzyme acetylcholinesterase, leading to increased levels of acetylcholine in the eye. This results in prolonged miosis and reduced intraocular pressure.
Mechanism of action[edit]
Demecarium bromide functions by inhibiting the breakdown of acetylcholine, a neurotransmitter involved in the contraction of the ciliary muscle of the eye. By preventing the degradation of acetylcholine, demecarium bromide enhances the outflow of aqueous humor through the trabecular meshwork, thereby lowering intraocular pressure. This mechanism is particularly beneficial in the treatment of open-angle glaucoma.
Clinical uses[edit]
Demecarium bromide is primarily indicated for the treatment of chronic open-angle glaucoma and secondary glaucoma. It is also used in the management of accommodative esotropia, a condition where the eyes turn inward due to excessive accommodation. The drug is administered as an ophthalmic solution, typically in concentrations ranging from 0.125% to 0.25%.
Side effects[edit]
Common side effects of demecarium bromide include ocular irritation, conjunctival hyperemia, and lacrimation. Prolonged use may lead to cataract formation and retinal detachment. Systemic absorption can result in cholinergic crisis, characterized by symptoms such as bradycardia, hypotension, and respiratory depression.
Contraindications[edit]
Demecarium bromide is contraindicated in patients with narrow-angle glaucoma, iritis, or uveitis. It should be used with caution in individuals with a history of asthma, epilepsy, or cardiovascular disease.
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian