Cinoxacin
Cinoxacin[edit]
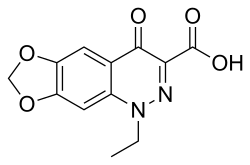
Cinoxacin is a synthetic antibiotic belonging to the class of quinolone antibiotics. It was primarily used to treat urinary tract infections (UTIs) caused by susceptible strains of bacteria. Cinoxacin was one of the earlier quinolones developed and was introduced in the 1970s.
Chemical Properties[edit]
Cinoxacin is a naphthyridine derivative, which is structurally related to other quinolones. Its chemical formula is C12H10N2O5, and it has a molecular weight of 262.22 g/mol. The compound is characterized by a bicyclic structure that includes a carboxylic acid group, which is essential for its antibacterial activity.
Mechanism of Action[edit]
Cinoxacin works by inhibiting bacterial DNA gyrase and topoisomerase IV, enzymes that are critical for DNA replication and transcription. By interfering with these enzymes, cinoxacin prevents the bacteria from replicating and repairing their DNA, leading to bacterial cell death.
Clinical Use[edit]
Cinoxacin was primarily used to treat uncomplicated urinary tract infections. It was effective against a range of Gram-negative bacteria, including Escherichia coli, Proteus mirabilis, and Klebsiella pneumoniae. However, its use has declined with the development of newer quinolones that have broader spectra of activity and improved pharmacokinetic properties.
Pharmacokinetics[edit]
Cinoxacin is well absorbed from the gastrointestinal tract after oral administration. It is widely distributed in body tissues and fluids, including the kidneys and urinary tract, which makes it effective for treating UTIs. The drug is primarily excreted unchanged in the urine.
Side Effects[edit]
Common side effects of cinoxacin include gastrointestinal disturbances such as nausea, vomiting, and diarrhea. Some patients may experience central nervous system effects like dizziness and headache. As with other quinolones, there is a risk of tendonitis and tendon rupture, although this is more common with newer agents.
Resistance[edit]
Bacterial resistance to cinoxacin can occur through mutations in the genes encoding DNA gyrase and topoisomerase IV, or through the acquisition of resistance genes via plasmids. The development of resistance has limited the clinical utility of cinoxacin and other early quinolones.
Discontinuation[edit]
Cinoxacin is no longer widely used in clinical practice, having been largely replaced by newer quinolones with improved efficacy and safety profiles. The availability of more potent and less toxic alternatives has led to a decline in its use.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian