Carbon-12
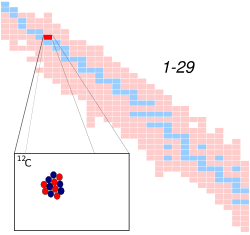
Carbon-12 is an isotope of carbon, denoted as 12
C
. It is the most abundant of the two stable isotopes of carbon, making up about 98.93% of the carbon found on Earth. The nucleus of a carbon-12 atom consists of 6 protons and 6 neutrons.
Properties[edit]
Carbon-12 has an atomic mass of exactly 12 amu by definition. This isotope is used as the standard from which atomic masses of all nuclides are measured, a decision made by the International Union of Pure and Applied Chemistry (IUPAC) in 1961.
Role in Science[edit]
Carbon-12 plays a crucial role in chemistry and physics. It is the basis for the mole, a fundamental unit in the International System of Units (SI) used to measure the amount of substance. One mole of carbon-12 atoms has a mass of exactly 12 grams.
Formation[edit]
Carbon-12 is formed in stars through the triple-alpha process, a set of nuclear fusion reactions by which three helium-4 nuclei (alpha particles) are transformed into carbon.
Applications[edit]
Carbon-12 is widely used in mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy due to its non-radioactive nature and its abundance. It is also used in carbon dating to determine the age of archaeological and geological samples.
Related Isotopes[edit]
Other isotopes of carbon include carbon-13 and carbon-14. Carbon-13 is a stable isotope used in isotopic labeling and NMR spectroscopy, while carbon-14 is a radioactive isotope used in radiocarbon dating.
See Also[edit]
- Isotope
- Carbon
- Atomic mass unit
- Mole (unit)
- Triple-alpha process
- Mass spectrometry
- Nuclear magnetic resonance
- Carbon-13
- Carbon-14
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian