Avacopan
What is Avacopan?[edit]
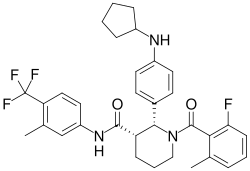
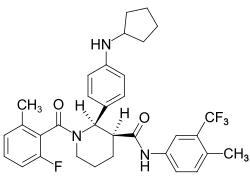
- Avacopan (Tavneos) is a complement 5a receptor (C5aR) antagonist used to treat severe active anti-neutrophil cytoplasmic autoantibody-associated vasculitis (granulomatosis with polyangiitis and microscopic polyangiitis) in combination with standard therapy, including glucocorticoids.
What are the uses of this medicine?[edit]
- Avacopan (Tavneos) is a prescription medicine that is used with other medicines (such as glucocorticoids) to treat adults with severe active anti-neutrophil cytoplasmic autoantibody (ANCA)- associated vasculitis (granulomatosis with polyangiitis [GPA], formerly known as Wegener’s granulomatosis, and microscopic polyangiitis [MPA]).
How does this medicine work?[edit]
- Avacopan is a complement 5a receptor (C5aR) antagonist that inhibits the interaction between C5aR and the anaphylatoxin C5a.
- Avacopan blocks C5a-mediated neutrophil activation and migration.
- The precise mechanism by which avacopan exerts a therapeutic effect in patients with ANCA-associated vasculitis has not been definitively established.
Who Should Not Use this medicine ?[edit]
This medicine cannot be used in patients with:
- serious hypersensitivity reaction to avacopan or to any of the excipients.
What drug interactions can this medicine cause?[edit]
- Avoid coadministration of strong and moderate CYP3A4 inducers with Tavneos.
- Avacopan exposure is increased when co-administered with strong CYP3A4 enzyme inhibitors such as itraconazole. Administer Tavneos 30 mg once daily when coadministered with strong CYP3A4 inhibitors.
- Closely monitor patients for adverse reactions and consider dose reduction of sensitive CYP3A4 substrates with a narrow therapeutic window when coadministered with Tavneos.
Is this medicine FDA approved?[edit]
- Avacopan was approved for medical use in Japan in September 2021, and in the United States in October 2021.
How should this medicine be used?[edit]
Before initiating Tavneos, consider performing the following evaluations:
- Liver Function Tests
- Hepatitis B (HBV) Serology
Recommended dosage:
- The recommended dosage is 30 mg (three 10 mg capsules twice daily with food).
Dosage Modifications Due to CYP3A4 Inhibitors:
- Reduce the dosage of Tavneos to 30 mg once daily when used concomitantly with strong CYP3A4 inhibitors.
Administration:
- Take 3 capsules of Tavneos 2 times daily (morning and evening) with food.
- Your healthcare provider may tell you to take 3 capsules of Tavneos 1 time each day if you take certain medicines. Tell your healthcare provider about all the medicines you take.
- Swallow the capsules whole with water. Do not crush, chew or open the capsules.
- If you miss a dose of Tavneos, do not take the missed dose. Just take the next dose at your regular time.
- If you have taken too much Tavneos, call your doctor or a Poison Control Center, or go to the nearest hospital emergency room.
What are the dosage forms and brand names of this medicine?[edit]
This medicine is available in fallowing doasage form:
- As Capsules: 10 mg
This medicine is available in fallowing brand namesː
- Tavneos
What side effects can this medication cause?[edit]
The most common side effects of this medicine include:
- nausea
- diarrhea
- tiredness
- increase in blood creatinine
- headache
- vomiting
- stomach pain
- burning or prickling sensation
- high blood pressure
- rash
- dizziness
Tavneos may cause serious side effects, including:
- Liver problems
- Serious allergic reactions
- Hepatitis B virus (HBV) reactivation
- Serious infections
What special precautions should I follow?[edit]
- Serious cases of hepatic injury have been observed in patients taking Tavneos. Increase in liver function tests occurred in clinical trials. Obtain liver function tests before initiation of therapy and monitor as clinically indicated.
- Tavneos may cause angioedema. Cases of angioedema occurred in a clinical trial. Observe for signs and symptoms of angioedema and manage accordingly.
- Hepatitis B virus (HBV) reactivation, including life threatening hepatitis B, was observed in the clinical program. Withhold Tavneos and institute appropriate anti-infective therapy.
- Serious infections, including fatal infections, have been reported in patients receiving Tavneos. Avoid use of Tavneos in patients with active, serious infection, including localized infections.
What to do in case of emergency/overdose?[edit]
- In case of overdose, call the poison control helpline of your country. In the United States, call 1-800-222-1222.
- Overdose related information is also available online at poisonhelp.org/help.
- In the event that the victim has collapsed, had a seizure, has trouble breathing, or can't be awakened, immediately call emergency services. In the United States, call 911.
Can this medicine be used in pregnancy?[edit]
- There are no adequate and well-controlled studies with Tavneos in pregnant women to inform a drug-associated risk.
- It is not known if Tavneos will harm your unborn baby.
Can this medicine be used in children?[edit]
- The safety and effectiveness of Tavneos in pediatric patients have not been established.
What are the active and inactive ingredients in this medicine?[edit]
- Active ingredient: avacopan
- Inactive ingredients: Polyethylene glycol 4000 (PEG-4000), Polyoxyl-40 hydrogenated castor oil.
Who manufactures and distributes this medicine?[edit]
Tavneos is a trademark of ChemoCentryx, Inc.
Manufactured for ChemoCentryx, Inc. by:
- Thermo Fisher Scientific
- 2110 East Galbraith Road
- Cincinnati, OH USA
What should I know about storage and disposal of this medication?[edit]
- Store Tavneos capsules at room temperature between 68°F to 77°F (20°C to 25°).
- Keep Tavneos and all medicines out of reach of children.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian