Aqueous solution
Aqueous Solution[edit]
An aqueous solution is a solution in which the solvent is water. It is denoted by the symbol (aq) in chemical equations. A common example of an aqueous solution is sodium chloride (NaCl) dissolved in water, forming a solution of Na_ and Cl_ ions.
Properties[edit]
Aqueous solutions have unique properties due to the polar nature of water molecules. Water's polarity allows it to dissolve many ionic compounds and polar molecules. This makes water an excellent solvent for a wide range of substances.
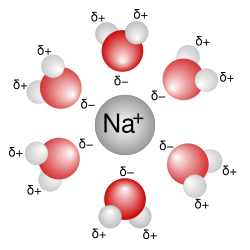
- Polarity: Water molecules have a partial negative charge near the oxygen atom and a partial positive charge near the hydrogen atoms, allowing them to interact with various solutes.
- Hydration: When ionic compounds dissolve in water, the ions become surrounded by water molecules, a process known as hydration.
- Conductivity: Aqueous solutions of ionic compounds can conduct electricity due to the presence of free-moving ions.
Types of Aqueous Solutions[edit]
Aqueous solutions can be classified based on the nature of the solute:
- Electrolytes: These are substances that dissolve in water to produce a solution that conducts electricity. Examples include sodium chloride and potassium nitrate.
- Non-electrolytes: These are substances that dissolve in water but do not conduct electricity. Examples include sugar and ethanol.
Chemical Reactions in Aqueous Solutions[edit]
Many important chemical reactions occur in aqueous solutions, including:
- Acid-base reactions: These involve the transfer of protons (H_) between reactants. For example, the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) in water.
- Precipitation reactions: These occur when two aqueous solutions are mixed and an insoluble solid, called a precipitate, forms.
- Redox reactions: These involve the transfer of electrons between species in solution.
Related Pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian