Anagestone acetate
An overview of Anagestone acetate, a synthetic progestogen
Anagestone acetate[edit]
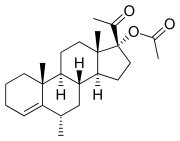
Anagestone acetate is a synthetic progestogen that belongs to the class of steroid hormones. It is used in hormonal contraception and hormone replacement therapy. Anagestone acetate is a derivative of progesterone, a natural hormone involved in the menstrual cycle, pregnancy, and embryogenesis.
Chemical properties[edit]
Anagestone acetate is a steroid with the chemical formula C24H32O4. It is a white to off-white crystalline powder that is practically insoluble in water but soluble in organic solvents. The compound is characterized by its acetate group, which is attached to the 17-alpha position of the steroid nucleus, enhancing its oral bioavailability.
Pharmacology[edit]
Anagestone acetate acts as a potent agonist of the progesterone receptor. By binding to this receptor, it mimics the effects of natural progesterone, leading to changes in the endometrium that prevent ovulation and alter the cervical mucus, making it more difficult for sperm to enter the uterus.
Mechanism of action[edit]
The primary mechanism of action of anagestone acetate is the suppression of the hypothalamic-pituitary-gonadal axis, which reduces the secretion of gonadotropins such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH). This suppression prevents the maturation of ovarian follicles and ovulation.
Clinical uses[edit]
Anagestone acetate is used in various hormonal therapies, including:
- Contraception: As a component of oral contraceptive pills, it helps prevent pregnancy.
- Hormone replacement therapy: Used to alleviate symptoms of menopause such as hot flashes and osteoporosis.
Side effects[edit]
Common side effects of anagestone acetate include:
Related pages[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian