Ammonium

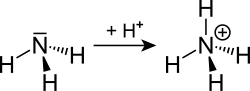

Ammonium (chemical formula: NH4+) is a positively charged polyatomic ion formed by the addition of a proton (H+) to an ammonia (NH3) molecule. It is a key component in the nitrogen cycle and plays a crucial role in the biochemistry of aquatic and terrestrial life forms. Ammonium ions are found in a variety of salts such as ammonium chloride (NH4Cl), ammonium nitrate (NH4NO3), and ammonium sulfate ((NH4)2SO4), which are widely used in fertilizers, explosives, and in various industrial processes.
Properties and Structure[edit]
The ammonium ion has a tetrahedral shape, similar to methane (CH4). This structure is due to the sp3 hybridization of the nitrogen atom in ammonia before the addition of the extra proton. The ion carries a +1 charge, distributed over the four hydrogen atoms and the nitrogen atom, making it a relatively stable ion under normal conditions.
Production[edit]
Ammonium ions are produced naturally in the soil by the decomposition of organic matter and the excretion of nitrogenous waste by animals. Industrially, ammonium salts are produced by reacting ammonia with various acids. For example, ammonium nitrate is produced by the reaction of ammonia with nitric acid, and ammonium sulfate is produced by reacting ammonia with sulfuric acid.
Applications[edit]
Fertilizers[edit]
Ammonium-based compounds are key ingredients in fertilizers, providing a readily available source of nitrogen to plants, which is essential for their growth and development. The use of ammonium nitrate and ammonium sulfate as fertilizers helps to increase crop yields and improve food production globally.
Explosives[edit]
Ammonium nitrate is a major component in many explosive mixtures used in mining, construction, and military applications due to its high nitrogen content and ability to release a large amount of energy upon decomposition.
Other Uses[edit]
Ammonium salts are also used in various industrial processes, including the production of plastics, dyes, and pharmaceuticals. They serve as flame retardants, fermentation nutrients in the production of antibiotics, and as a component in household cleaning products.
Environmental Impact[edit]
While ammonium ions play a vital role in the nitrogen cycle, excessive use of ammonium-based fertilizers can lead to environmental issues such as eutrophication of water bodies and soil acidification. The leaching of ammonium into waterways can stimulate the growth of algae and other aquatic plants, leading to decreased oxygen levels and the death of aquatic organisms.
Health Effects[edit]
In humans, exposure to high concentrations of ammonium compounds, especially in their gaseous form (ammonia), can cause irritation to the eyes, nose, and throat, and in severe cases, respiratory distress. However, ammonium ions present in drinking water or food are generally not harmful at low concentrations.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
