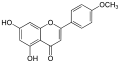
Acacetin
A flavone compound found in various plants
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Acacetin is a naturally occurring flavone found in various plant species. It is known for its potential biological activity and is studied for its effects on human health.
Structure and Properties[edit]
Acacetin is a type of flavonoid, specifically a flavone, characterized by its chemical structure which includes a phenyl group attached to a chromen-4-one backbone. The molecular formula of acacetin is C16H12O5, and it has a molecular weight of 284.26 g/mol.
Occurrence[edit]
Acacetin is found in several plant species, including Robinia pseudoacacia (black locust) and Turnera diffusa (damiana). It is often present in the leaves and flowers of these plants.
Biological Activity[edit]
Acacetin has been studied for its potential antioxidant, anti-inflammatory, and anticancer properties. It is believed to exert these effects by modulating various cellular pathways and enzyme activities.
Antioxidant Properties[edit]
As an antioxidant, acacetin can scavenge free radicals and reduce oxidative stress in cells. This activity is important in protecting cells from damage and may contribute to its potential health benefits.
Anti-inflammatory Effects[edit]
Acacetin has been shown to inhibit the production of pro-inflammatory cytokines and reduce inflammation in various in vitro and in vivo models. This makes it a compound of interest in the study of inflammatory diseases.
Anticancer Potential[edit]
Research suggests that acacetin may have anticancer properties by inducing apoptosis in cancer cells and inhibiting their proliferation. It has been studied in the context of various types of cancer, including breast cancer and prostate cancer.
Synthesis[edit]
Acacetin can be synthesized through various chemical methods, often involving the hydroxylation of flavone precursors. These synthetic methods are important for producing acacetin in sufficient quantities for research and potential therapeutic use.
Uses[edit]
While acacetin is primarily of interest in research settings, it is also explored for its potential use in nutraceuticals and pharmaceuticals. Its natural occurrence in plants also makes it a component of some traditional herbal remedies.
Related Pages[edit]
Gallery[edit]
-
Skeletal formula of acacetin
-
Ball-and-stick model of acacetin
Acacetin[edit]
-
Acacetin chemical structure
-
Acacetin molecule ball-and-stick model
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian