Radical (chemistry)
Radical (chemistry)
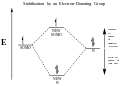
A radical in chemistry is an atom, molecule, or ion that has unpaired valence electrons. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes.
A notable example of a radical is the hydroxyl radical (HO•), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and benzyl radical.
Formation and occurrence of radicals[edit]
Radicals can be formed by ionizing radiation, heat, electrical discharge, or photochemical reactions. Certain radicals are used in radical polymerization and radical chain reactions, which are essential to many processes in chemistry, biochemistry, and medicine.
Reactivity and applications of radicals[edit]
Radicals play key roles in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes, including human physiology. For example, in living organisms, radicals are involved in cell damage and cancer.
See also[edit]
- Radical
- Radical polymerization
- Radical chain reaction
- Hydroxyl radical
- Triplet oxygen
- Benzyl radical
References[edit]
Radical (chemistry)[edit]
-
Hydroxyl radical
-
Hydroxide vs hydroxyl
-
Bromine homolysis
-
Homolysis
-
Lithium Naphthalenide
-
Abstraction
-
Radical addition
-
Radical elimination
-
2,2,6,6-Tetramethylpiperidinyloxyl
-
Electron-Donating Diagram
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian