4-Fluorophenibut
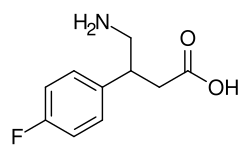
4-Fluorophenibut is a GABAergic compound and a derivative of phenibut, a neurotransmitter-modulating drug. It is chemically similar to phenibut but with the addition of a fluorine atom at the para position of the phenyl ring. This modification is believed to affect its pharmacological properties, potentially making it more potent or altering its bioavailability and metabolism. 4-Fluorophenibut acts primarily as a GABA_B receptor agonist, similar to phenibut, but it may also have some activity at GABA_A receptors. Its effects on the central nervous system (CNS) include anxiolytic, euphoric, nootropic, and sedative properties.
Pharmacology[edit]
The pharmacological profile of 4-Fluorophenibut is characterized by its action on GABA receptors, the primary inhibitory neurotransmitter system in the brain. By mimicking GABA, it can induce a calming effect on the CNS. The addition of a fluorine atom may enhance its penetration through the blood-brain barrier, potentially increasing its effectiveness compared to phenibut.
Clinical Uses[edit]
As of now, 4-Fluorophenibut has not been approved for any clinical use in most countries. It is primarily researched in academic or pharmaceutical settings to understand its pharmacodynamics and pharmacokinetics. Its potential therapeutic applications could mirror those of phenibut, including the treatment of anxiety, insomnia, and various other CNS disorders. However, without formal clinical trials, these uses remain speculative.
Safety and Side Effects[edit]
The safety profile of 4-Florophenibut is not well-documented, given its status as a research chemical rather than a clinically approved drug. Potential side effects may resemble those of phenibut, including dizziness, nausea, sedation, and the risk of dependence with prolonged use. The addition of the fluorine atom could also introduce unique toxicological properties not present in phenibut, emphasizing the need for cautious use and further research.
Legal Status[edit]
The legal status of 4-Fluorophenibut varies by country, with some jurisdictions classifying it as a controlled substance due to its structural similarity to phenibut and potential for abuse. In countries where it is not explicitly controlled, it may still fall under analogue acts or be subject to restrictions on psychoactive substances.
Conclusion[edit]
4-Fluorophenibut represents an interesting area of research in the field of psychoactive substances, offering potential insights into the modulation of the GABAergic system. However, its use outside of controlled research settings is not recommended due to the lack of comprehensive safety data and its uncertain legal status.
|
This article is a stub You can help WikiMD by registering and expanding it with useful details, internal links, formatting, and categories. Editing is available only to registered and verified users. WikiMD is a comprehensive, free health and wellness encyclopedia. |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
