2-Arachidonoylglycerol
2-Arachidonoylglycerol[edit]
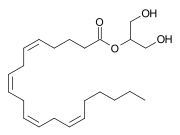
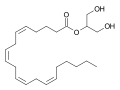
2-Arachidonoylglycerol (2-AG) is an endocannabinoid, an endogenous eicosanoid molecule that binds to the cannabinoid receptors. It is an ester formed from the omega-6 fatty acid arachidonic acid and glycerol.
Structure[edit]
2-Arachidonoylglycerol is a glycerol ester of arachidonic acid, a polyunsaturated fatty acid with a 20-carbon chain and four cis-double bonds. The chemical formula of 2-AG is C23H38O4.
Biosynthesis[edit]
2-AG is synthesized in the body from phospholipids in the cell membrane. The primary pathway involves the hydrolysis of diacylglycerol (DAG) by the enzyme diacylglycerol lipase (DAGL). This process releases 2-AG as a free molecule.
Function[edit]
2-Arachidonoylglycerol acts as a full agonist at both the CB1 and CB2 cannabinoid receptors. These receptors are part of the endocannabinoid system, which plays a role in regulating various physiological processes including appetite, pain sensation, mood, and memory.
Metabolism[edit]
2-AG is metabolized primarily by the enzyme monoacylglycerol lipase (MAGL), which hydrolyzes it to arachidonic acid and glycerol. Other enzymes, such as fatty acid amide hydrolase (FAAH), can also contribute to its breakdown.
Clinical Significance[edit]
Alterations in 2-AG levels have been associated with various pathological conditions, including neurodegenerative diseases, inflammation, and cancer. Research is ongoing to explore the therapeutic potential of modulating 2-AG levels in these conditions.
Related pages[edit]
Gallery[edit]
-
Chemical structure of 2-Arachidonoylglycerol
-
3D model of 2-Arachidonoylglycerol
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian