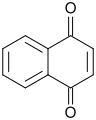
1,4-Naphthoquinone
1,4-Naphthoquinone[edit]
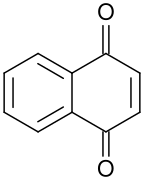
1,4-Naphthoquinone is an organic compound with the formula C__H_O_. It is a yellow crystalline solid that is a derivative of naphthalene. It is one of the two isomeric naphthoquinones, the other being 1,2-naphthoquinone.
Structure and Properties[edit]
1,4-Naphthoquinone consists of a naphthalene ring with two ketone groups at the 1 and 4 positions. This structure imparts a planar geometry to the molecule, which is typical of quinones. The compound is characterized by its yellow color and its ability to undergo redox reactions, making it a useful intermediate in various chemical processes.
Synthesis[edit]
1,4-Naphthoquinone can be synthesized through the oxidation of naphthalene using various oxidizing agents such as chromic acid or potassium permanganate. Another method involves the oxidation of 1,4-dihydroxynaphthalene.
Reactions[edit]
1,4-Naphthoquinone is a versatile compound in organic synthesis. It can undergo Diels-Alder reactions with dienes, such as butadiene, to form adducts. This reaction is useful in the synthesis of complex organic molecules.
Applications[edit]
1,4-Naphthoquinone is used in the production of dyes, vitamin K analogs, and as a precursor to various pharmaceuticals. Its ability to participate in redox reactions makes it valuable in the study of electron transfer processes.
Safety[edit]
1,4-Naphthoquinone is a hazardous substance and should be handled with care. It can cause irritation to the skin, eyes, and respiratory tract. Proper safety precautions, including the use of personal protective equipment, should be observed when handling this compound.
Related Compounds[edit]
Related Pages[edit]
-
Structural formula of 1,4-Naphthoquinone
-
3D ball-and-stick model of 1,4-Naphthoquinone
-
Reaction of 1,4-Naphthoquinone with butadiene
- 1,4-Naphthoquinone
-
1,4-Naphthoquinone
-
1,4-Naphthoquinone 3D model
-
Naphthoquinone reaction with butadiene
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian