Potassium permanganate
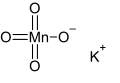
Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a salt consisting of K+ and MnO4- ions. It is a strong oxidizing agent. The salt is also known as "permanganate of potash" and "Condy's crystals". In this salt, manganese is in the +7 oxidation state.
History[edit]
The compound was first developed in 1659 by the German chemist Johann Rudolf Glauber. It was not until 1857 that Henry Bollmann Condy began to market the compound as a disinfectant.
Properties[edit]
Potassium permanganate is a purplish-black crystalline solid, that dissolves in water to give intensely pink or purple solutions. Its melting point is 240 °C (464 °F), and it decomposes at higher temperatures.
Uses[edit]
Potassium permanganate is used for a number of skin conditions. This includes fungal infections of the foot, impetigo, pemphigus, superficial wounds, dermatitis, and tropical ulcers. It is on the World Health Organization's List of Essential Medicines, the safest and most effective medicines needed in a health system.
Safety[edit]
Potassium permanganate is a strong oxidizer and can cause burns and ulcers in concentrated form. It is harmful if swallowed and can cause severe gastrointestinal effects.
See also[edit]
References[edit]
Potassium_permanganate[edit]
-
KMnO4.svg
-
Potassium-permanganate-2004-unit-cell-3D-balls.png
-
Potassium-permanganate-sample.jpg
-
KMnO4 in H2O.jpg
-
Baeyer's Probe Alkenes V.2.svg
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian