1,2-Dimethoxybenzene
1,2-Dimethoxybenzene, also known as veratrole, is an organic compound that is commonly used in organic synthesis. It is a derivative of benzene, containing two methoxy functional groups.
Structure and Properties[edit]
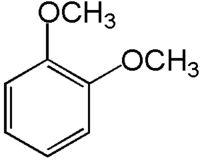
1,2-Dimethoxybenzene is a colorless liquid that is slightly soluble in water but highly soluble in organic solvents. It has a pleasant odor and is used in perfumery and as a flavoring agent. The compound has a molecular formula of C8H10O2 and a molecular weight of 138.16 g/mol. Its structure consists of a benzene ring with two methoxy groups attached at the 1 and 2 positions.
Synthesis[edit]
1,2-Dimethoxybenzene can be synthesized by the methylation of guaiacol using dimethyl sulfate or methyl iodide in the presence of a base. The reaction proceeds via an SN2 reaction mechanism.
Applications[edit]
1,2-Dimethoxybenzene is used as a building block in organic synthesis. It is also used in the production of perfumes and flavorings due to its pleasant odor. In addition, it is used as a solvent for chemical reactions and in the manufacture of dyes and pharmaceuticals.
Safety[edit]
1,2-Dimethoxybenzene is harmful if swallowed, inhaled, or comes into contact with the skin. It can cause irritation to the eyes, skin, and respiratory tract. It is recommended to use personal protective equipment when handling this compound.
See Also[edit]
References[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
