DNA glycosylase
DNA glycosylases are a family of enzymes that play a crucial role in the repair of DNA by initiating the base excision repair (BER) pathway. These enzymes recognize and remove damaged or inappropriate bases from DNA, thereby preventing mutations that could lead to cancer, aging, and various genetic disorders. DNA glycosylases have been identified in a wide range of organisms, from bacteria to humans, highlighting their importance in maintaining genomic stability.
Function[edit]
DNA glycosylases function by flipping the damaged base out of the DNA helix and into the enzyme's active site. Once the damaged base is removed, the site is left apurinic/apyrimidinic (AP site), which is then recognized and processed by other enzymes in the BER pathway. This process involves several steps, including the removal of the sugar-phosphate backbone, filling in the gap with the correct base, and sealing the nick in the DNA backbone. The specificity of DNA glycosylases for different types of damage is a key aspect of their function, with different enzymes recognizing specific types of damaged bases, such as those caused by oxidative stress, alkylation, or deamination.
Types of DNA Glycosylases[edit]
There are several types of DNA glycosylases, each with specificity for different damaged or inappropriate bases. Some of the well-known DNA glycosylases include:
- Uracil-DNA glycosylase (UNG): Recognizes and removes uracil from DNA, which can arise through deamination of cytosine or incorporation during DNA replication.
- 8-Oxoguanine DNA glycosylase (OGG1): Targets 8-oxoguanine, a common lesion formed by oxidative damage, which can mispair with adenine during replication.
- Thymine DNA glycosylase (TDG): Removes thymine glycol and other oxidative thymine lesions, as well as thymine mispaired with guanine.
- MutY DNA glycosylase (MUTYH): Removes adenines misincorporated opposite 8-oxoguanine, preventing G:C to T:A transversion mutations.
Clinical Significance[edit]
Mutations in the genes encoding DNA glycosylases can lead to a predisposition to cancer and other diseases. For example, mutations in the MUTYH gene are associated with MUTYH-associated polyposis (MAP), a condition that increases the risk of developing colorectal cancer. Understanding the mechanisms of action and regulation of DNA glycosylases is crucial for developing novel therapeutic strategies for treating diseases associated with DNA damage and repair defects.
Research and Therapeutic Applications[edit]
Research into DNA glycosylases has led to the development of inhibitors and synthetic lethality strategies for cancer therapy. By targeting specific DNA repair pathways in cancer cells, which often rely on these mechanisms due to their high rate of DNA damage, it is possible to selectively kill cancer cells while sparing normal cells. Additionally, understanding the role of DNA glycosylases in aging and neurodegenerative diseases could lead to the development of interventions aimed at promoting genomic stability and healthy aging.
See Also[edit]

This article is a molecular biology stub. You can help WikiMD by expanding it!
-
Uracil base glycosidase
-
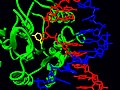
PDB structure 2j8x
-
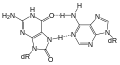
8-oxoG forming Hoogsteen base pair with dA
-
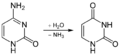
Deamination of cytosine to uracil
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian