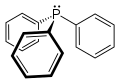Triphenylphosphine
An organophosphorus compound used in organic synthesis
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Triphenylphosphine (often abbreviated as TPP or PPh_) is an organophosphorus compound with the formula P(C_H_)_. It is a common reagent in organic synthesis and is used as a ligand in coordination chemistry.
Structure and properties[edit]
Triphenylphosphine is a white crystalline solid at room temperature. It is soluble in organic solvents such as benzene, diethyl ether, and chloroform. The compound is characterized by a trigonal pyramidal geometry around the phosphorus atom, with three phenyl groups attached.
Synthesis[edit]
Triphenylphosphine is typically synthesized by the reaction of phosphorus trichloride with phenylmagnesium bromide or phenyl lithium. The reaction proceeds as follows:
Applications[edit]
In organic synthesis[edit]
Triphenylphosphine is widely used in organic synthesis. It is a key component in the Wittig reaction, which is used to convert carbonyl compounds into alkenes. The reaction involves the formation of a phosphonium ylide intermediate.
Deoxygenation reactions[edit]
Triphenylphosphine is also used in deoxygenation reactions, such as the conversion of amine oxides to amines. This reaction is depicted in the following image:

As a ligand[edit]
In coordination chemistry, triphenylphosphine acts as a ligand that can stabilize transition metal complexes. It is often used in homogeneous catalysis, such as in the hydroformylation of olefins.
Derivatives[edit]
Triphenylphosphine can be modified to form various derivatives, such as TPPTS (triphenylphosphine-3,3',3-trisulfonate), which is used in aqueous catalysis.

Safety[edit]
Triphenylphosphine is generally considered to be of low toxicity, but it can cause irritation to the skin and eyes. Proper handling and storage are recommended to avoid exposure.
Related pages[edit]
References[edit]
-
Triphenylphosphine 2D skeletal structure
-
Deoxygenation of an aromatic amine oxide using triphenylphosphine
-
TPPTS
Ad. Transform your health with W8MD Weight Loss, Sleep & MedSpa

Tired of being overweight?
Special offer:
Budget GLP-1 weight loss medications
- Semaglutide starting from $29.99/week and up with insurance for visit of $59.99 and up per week self pay.
- Tirzepatide starting from $45.00/week and up (dose dependent) or $69.99/week and up self pay
✔ Same-week appointments, evenings & weekends
Learn more:
- GLP-1 weight loss clinic NYC
- W8MD's NYC medical weight loss
- W8MD Philadelphia GLP-1 shots
- Philadelphia GLP-1 injections
- Affordable GLP-1 shots NYC
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian