Gibbs–Donnan effect
Gibbs–Donnan Effect[edit]
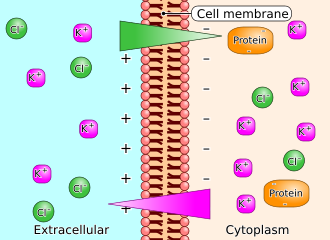
The Gibbs–Donnan effect, also known as the Donnan effect, describes the behavior of charged particles near a semi-permeable membrane that does not allow certain ions to pass through. This phenomenon is named after the American physicist Josiah Willard Gibbs and the British chemist Frederick George Donnan.
Principles[edit]
The Gibbs–Donnan effect occurs when a semi-permeable membrane separates two solutions, one of which contains a non-diffusible ion. This situation leads to an unequal distribution of diffusible ions across the membrane, resulting in an osmotic pressure difference and an electric potential difference between the two sides.
Ion Distribution[edit]
In a typical scenario, consider a membrane that separates two compartments. One compartment contains a solution with a non-diffusible anion, such as a large protein, while the other compartment contains a simple salt solution. The presence of the non-diffusible anion causes an imbalance in the distribution of diffusible ions, such as sodium (Na_) and chloride (Cl_) ions, across the membrane.
Osmotic Pressure[edit]
The unequal distribution of ions results in an osmotic pressure difference. Water tends to move from the side with lower osmotic pressure to the side with higher osmotic pressure, which can lead to swelling or shrinkage of cells or compartments.
Electric Potential[edit]
The Gibbs–Donnan effect also creates an electric potential difference across the membrane. This potential difference is due to the unequal distribution of charged ions, which can affect the movement of other ions and molecules across the membrane.
Biological Significance[edit]
The Gibbs–Donnan effect is important in various biological systems. It plays a crucial role in maintaining the cell membrane potential and the distribution of ions across cell membranes. In particular, it affects the behavior of red blood cells, neurons, and other cells that rely on ion gradients for their function.
Red Blood Cells[edit]
In red blood cells, the Gibbs–Donnan effect contributes to the maintenance of cell volume and the balance of ions such as potassium (K_) and sodium (Na_) across the cell membrane.
Neurons[edit]
In neurons, the effect is part of the mechanism that establishes the resting membrane potential, which is essential for the transmission of nerve impulses.
Related Pages[edit]
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian