Osmosis
Osmosis is the movement of water or other solvent through a plasma membrane from a region of low solute concentration to a region of high solute concentration.[1] Osmosis is passive transport, meaning it does not require energy to be applied. What causes osmotic pressure is different concentrations of solutes on the two sides of the membrane.
When osmosis happens, water moves from the side of the membrane with a lower amount of osmotic pressure to the side of the membrane with the higher amount. An important example of osmosis is the movement of liquid (solvent) molecules across a cell membrane into a cell with a higher solute concentration. Osmosis is a fundamental part of cell biochemistry, but also has mechanical applications and usages.
Hypotonic, isotonic, and hypertonic[edit]
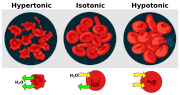
Solutions may have more or less solute per unit of solvent. The one with less is called Hypotonic. When the two solutions have equal concentration, they are isotonic. The one with more is Hypertonic. When the hypotonic solution is outside the cell,and hypertonic solution inside, the cell becomes swollen and distorted.
Cell membranes[edit]
The plasma membrane of a cell is semi-permeable, which means it allowed the entry of certain molecules in or out, it lets small molecules pass through, but blocks larger molecules,. The membrane also has ports or gateways which get certain macromolecules through. This is active transport, which uses energy and is selective. It is outermost covering of the animal cell. It is made up of proteins and lipids. Example:- Exchange of gases like oxygen and carbon dioxide.
Related pages[edit]
- ↑ http://physioweb.med.uvm.edu/bodyfluids/osmosis.htm University of Vermont, Department of Physiology
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian