Alcohol (chemistry)


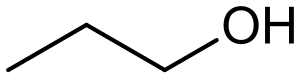


Alcohol (chemistry) refers to a class of organic compounds characterized by the presence of one or more hydroxyl groups (-OH) attached to a carbon atom. Alcohols are widely used in industry and everyday life, from the production of pharmaceuticals and cosmetics to being an active ingredient in alcoholic beverages. The structure and properties of an alcohol depend on the number of hydroxyl groups present and their attachment to the carbon chain.
Types of Alcohols[edit]
Alcohols can be classified into three main types based on the carbon atom to which the hydroxyl group is attached:
- Primary Alcohols (1°): The hydroxyl group is attached to a carbon atom that is bonded to only one other carbon atom. An example is ethanol, commonly found in alcoholic beverages.
- Secondary Alcohols (2°): The hydroxyl group is attached to a carbon atom that is bonded to two other carbon atoms. An example is isopropanol, used as a disinfectant.
- Tertiary Alcohols (3°): The hydroxyl group is attached to a carbon atom that is bonded to three other carbon atoms. An example is tert-butyl alcohol, used as a solvent and in organic synthesis.
Properties[edit]
Alcohols exhibit both hydrophilic and hydrophobic properties, making them good solvents for a wide range of substances. The boiling points of alcohols are generally higher than those of alkanes of similar molecular weight, due to hydrogen bonding between the hydroxyl groups. Alcohols can also undergo a variety of chemical reactions, including dehydration, oxidation, and esterification.
Uses[edit]
Alcohols have a wide range of applications:
- As solvents in the pharmaceutical, cosmetics, and paint industries.
- In the production of fuels, such as ethanol used in E85 gasoline.
- As disinfectants, such as isopropanol and ethanol in hand sanitizers.
- In the manufacture of plastics, perfumes, and other chemicals.
Health and Safety[edit]
While some alcohols, like ethanol, are safe in small quantities, others can be toxic or even lethal. Methanol, for example, is highly toxic and can cause blindness or death if ingested. Proper handling and storage of alcohols are essential to prevent accidents and health risks.
Environmental Impact[edit]
The production and use of alcohols, especially as fuels, have environmental implications. While bioethanol is considered a renewable energy source, its production and use can lead to deforestation, water use issues, and the emission of greenhouse gases. The environmental impact depends on the source of the alcohol and the methods used in its production and disposal.
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99


W8MD Medical Weight Loss, Sleep and Medspa offers physician-supervised medical weight loss programs: NYC medical weight loss Philadelphia medical weight loss
Affordable GLP-1 Weight Loss ShotsAffordable GLP-1 Weight Loss Shots
Budget GLP-1 injections NYC (insurance & self-pay options) Popular treatments:
- Semaglutide starting from $29.99/week
- Tirzepatide starting from $45.00/week
✔ Most insurances accepted for visits ✔ Prior authorization support when eligible
Start your physician weight loss NYC journey today:
📍 NYC: Brooklyn weight loss center 📍 Philadelphia: Philadelphia weight loss center
📞 Call: 718-946-5500 (NYC) | 215-676-2334 (Philadelphia)
Tags: Affordable GLP1 weight loss NYC, Wegovy NYC, Zepbound NYC, Philadelphia medical weight loss
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is not a substitute for professional medical advice. The information on WikiMD is provided as an information resource only, may be incorrect, outdated or misleading, and is not to be used or relied on for any diagnostic or treatment purposes. Please consult your health care provider before making any healthcare decisions or for guidance about a specific medical condition. WikiMD expressly disclaims responsibility, and shall have no liability, for any damages, loss, injury, or liability whatsoever suffered as a result of your reliance on the information contained in this site. By visiting this site you agree to the foregoing terms and conditions, which may from time to time be changed or supplemented by WikiMD. If you do not agree to the foregoing terms and conditions, you should not enter or use this site. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
