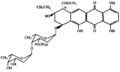
Bohemic acid
Bohemic acid is a chemical compound that belongs to the class of organic compounds known as phenols. It is a naturally occurring substance found in certain types of fungi, including species of the genus Bohemia.
Chemical Structure
Bohemic acid is a monocyclic compound with a single benzene ring in its structure. It has a molecular formula of C7H6O3 and a molecular weight of 138.12 g/mol. The compound is characterized by the presence of a hydroxyl group (-OH) attached to the benzene ring, which is responsible for its phenolic properties.
Properties
Bohemic acid is a white, crystalline solid at room temperature. It is soluble in water and most organic solvents, including ethanol, methanol, and acetone. The compound has a melting point of 132-134 °C and a boiling point of 310 °C. It is stable under normal conditions but can decompose upon exposure to light or heat, producing toxic fumes of carbon monoxide and carbon dioxide.
Biological Activity
Bohemic acid has been studied for its potential antimicrobial and antifungal activities. Some studies have shown that it can inhibit the growth of certain types of bacteria and fungi, including Staphylococcus aureus and Candida albicans. However, further research is needed to confirm these findings and to determine the exact mechanisms of action of bohemic acid.
Safety
As with all phenolic compounds, bohemic acid should be handled with care. It can cause irritation to the skin and eyes, and prolonged exposure can lead to serious health effects. Always use appropriate personal protective equipment when handling bohemic acid, and follow all safety guidelines and regulations.
See Also
Bohemic_acid
-
Alcindoromycin structure
-
Bohemanine structure
-
Musettamycin structure
-
Marcellomycin structure
-
Rudolphomycin structure
-
Schaunardimycin structure
Ad. Transform your life with W8MD's
GLP-1 weight loss injections special from $29.99 with insurance
|
WikiMD Medical Encyclopedia |
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian