Ethchlorvynol: Difference between revisions
CSV import |
CSV import |
||
| Line 1: | Line 1: | ||
{{Short description|Overview of the sedative and hypnotic drug Ethchlorvynol}} | |||
== | ==Ethchlorvynol== | ||
[[File:Ethchlorvynol_DOJ.jpg|thumb|right|Ethchlorvynol chemical structure]] | |||
'''Ethchlorvynol''' is a [[sedative]] and [[hypnotic]] medication that was primarily used for the treatment of [[insomnia]]. It was first introduced in the 1950s and marketed under the brand name Placidyl. Ethchlorvynol is a member of the [[pyrrolidine]] class of drugs and acts as a central nervous system [[depressant]]. | |||
Ethchlorvynol | ==Pharmacology== | ||
Ethchlorvynol works by enhancing the activity of the [[neurotransmitter]] [[gamma-aminobutyric acid]] (GABA) in the brain, which results in sedative and hypnotic effects. It is known to have a rapid onset of action, typically within 15 to 30 minutes, and its effects can last for several hours. The drug is metabolized in the liver and excreted by the kidneys. | |||
== Medical | ==Medical Uses== | ||
Ethchlorvynol was primarily prescribed for the short-term management of [[insomnia]], particularly in patients who had difficulty falling asleep. It was also occasionally used as a [[pre-anesthetic]] medication due to its sedative properties. | |||
==Side Effects== | |||
Common side effects of ethchlorvynol include [[drowsiness]], [[dizziness]], [[headache]], and [[nausea]]. In some cases, it can cause [[paradoxical reactions]] such as [[agitation]] or [[excitement]]. Long-term use can lead to [[tolerance]], [[dependence]], and [[withdrawal symptoms]] upon discontinuation. | |||
== | ==Synthesis== | ||
[[File:Ethchlorvynol_synthesis.png|thumb|left|Ethchlorvynol synthesis pathway]] | |||
The synthesis of ethchlorvynol involves the reaction of [[propargyl alcohol]] with [[trichloroethylene]] in the presence of a [[base]] to form the ethchlorvynol compound. This process highlights the chemical transformation necessary to produce the active sedative agent. | |||
==Regulation and Discontinuation== | |||
Due to concerns about its safety profile, including the risk of [[overdose]] and [[abuse]], ethchlorvynol was gradually phased out in many countries. In the United States, the manufacturer discontinued its production in 1999, and it is no longer available for prescription. | |||
== | |||
==Related Pages== | |||
* [[Sedative]] | * [[Sedative]] | ||
* [[Hypnotic]] | * [[Hypnotic]] | ||
* [[Insomnia]] | * [[Insomnia]] | ||
* [[ | * [[Gamma-aminobutyric acid]] | ||
* [[ | * [[Central nervous system depressant]] | ||
[[Category:Sedatives]] | [[Category:Sedatives]] | ||
[[Category:Hypnotics]] | [[Category:Hypnotics]] | ||
[[Category: | [[Category:Withdrawn drugs]] | ||
Latest revision as of 11:10, 23 March 2025
Overview of the sedative and hypnotic drug Ethchlorvynol
Ethchlorvynol[edit]
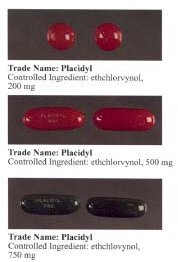
Ethchlorvynol is a sedative and hypnotic medication that was primarily used for the treatment of insomnia. It was first introduced in the 1950s and marketed under the brand name Placidyl. Ethchlorvynol is a member of the pyrrolidine class of drugs and acts as a central nervous system depressant.
Pharmacology[edit]
Ethchlorvynol works by enhancing the activity of the neurotransmitter gamma-aminobutyric acid (GABA) in the brain, which results in sedative and hypnotic effects. It is known to have a rapid onset of action, typically within 15 to 30 minutes, and its effects can last for several hours. The drug is metabolized in the liver and excreted by the kidneys.
Medical Uses[edit]
Ethchlorvynol was primarily prescribed for the short-term management of insomnia, particularly in patients who had difficulty falling asleep. It was also occasionally used as a pre-anesthetic medication due to its sedative properties.
Side Effects[edit]
Common side effects of ethchlorvynol include drowsiness, dizziness, headache, and nausea. In some cases, it can cause paradoxical reactions such as agitation or excitement. Long-term use can lead to tolerance, dependence, and withdrawal symptoms upon discontinuation.
Synthesis[edit]

The synthesis of ethchlorvynol involves the reaction of propargyl alcohol with trichloroethylene in the presence of a base to form the ethchlorvynol compound. This process highlights the chemical transformation necessary to produce the active sedative agent.
Regulation and Discontinuation[edit]
Due to concerns about its safety profile, including the risk of overdose and abuse, ethchlorvynol was gradually phased out in many countries. In the United States, the manufacturer discontinued its production in 1999, and it is no longer available for prescription.