Carisbamate: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 32: | Line 32: | ||
[[Category:Anticonvulsants]] | [[Category:Anticonvulsants]] | ||
[[Category:Carbamates]] | [[Category:Carbamates]] | ||
== Carisbamate == | |||
<gallery> | |||
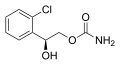
File:Carisbamate2DACS.svg|Carisbamate | |||
</gallery> | |||
Latest revision as of 01:37, 20 February 2025
An anticonvulsant drug candidate
| Carisbamate | |
|---|---|

| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Carisbamate is a drug candidate that has been investigated for its potential use as an anticonvulsant medication. It was developed by Johnson & Johnson and has been studied for the treatment of epilepsy and other neurological disorders.
Pharmacology[edit]
Carisbamate is believed to exert its effects by modulating neurotransmitter systems in the brain, although its precise mechanism of action is not fully understood. It has been shown to have activity in various animal models of epilepsy, suggesting its potential utility in managing seizures.
Clinical Trials[edit]
Carisbamate has undergone several clinical trials to evaluate its safety and efficacy. Initial studies indicated that it might be effective in reducing seizure frequency in patients with partial-onset seizures. However, subsequent trials did not demonstrate sufficient efficacy to warrant approval by regulatory agencies such as the FDA.
Development History[edit]
The development of carisbamate began in the early 2000s, with Johnson & Johnson conducting preclinical and clinical studies. Despite promising early results, the drug did not meet the necessary endpoints in later-stage trials, leading to a halt in its development for epilepsy.
Potential Uses[edit]
While carisbamate was primarily investigated for epilepsy, there is interest in exploring its effects on other neurological conditions, such as neuropathic pain and bipolar disorder. However, further research is needed to establish its efficacy and safety in these areas.
Chemical Properties[edit]
Carisbamate is a carbamate derivative, and its chemical structure is characterized by the presence of two carbamate groups. The molecular formula is C7H12N2O4, and it has a molecular weight of 188.18 g/mol.
Related pages[edit]
Carisbamate[edit]
-
Carisbamate