Velpatasvir: Difference between revisions
No edit summary |
CSV import |
||
| Line 1: | Line 1: | ||
{{Short description| | {{Short description|An antiviral medication used in the treatment of hepatitis C}} | ||
{{Drugbox | |||
| Verifiedfields = changed | |||
| verifiedrevid = 477002123 | |||
| image = Velpatasvir.svg | |||
| image2 = | |||
}} | |||
'''Velpatasvir''' is an [[antiviral drug]] used in combination with other medications for the treatment of [[hepatitis C virus]] (HCV) infection. It is a [[direct-acting antiviral]] agent that inhibits the [[NS5A protein]], which is essential for viral replication. | |||
Velpatasvir is used in combination with | |||
==Mechanism of | ==Mechanism of Action== | ||
Velpatasvir | Velpatasvir works by targeting the NS5A protein of the hepatitis C virus. NS5A is a non-structural protein that plays a crucial role in the replication of the virus and the assembly of the viral particle. By inhibiting NS5A, velpatasvir disrupts the replication cycle of HCV, thereby reducing the viral load in the body. | ||
==Side | ==Clinical Use== | ||
Common side effects of | Velpatasvir is commonly used in combination with [[sofosbuvir]], another antiviral medication, to form a fixed-dose combination known as [[sofosbuvir/velpatasvir]]. This combination is effective against all six major genotypes of hepatitis C, making it a [[pan-genotypic]] treatment option. It is typically administered as a once-daily oral tablet. | ||
==Side Effects== | |||
Common side effects of velpatasvir, especially when used in combination with sofosbuvir, include headache, fatigue, and nausea. Serious side effects are rare but may include [[bradycardia]] when used with [[amiodarone]]. | |||
==Pharmacokinetics== | ==Pharmacokinetics== | ||
Velpatasvir is well absorbed orally, with peak plasma concentrations occurring approximately 3 hours after dosing. It | Velpatasvir is well absorbed when taken orally, with peak plasma concentrations occurring approximately 3 hours after dosing. It is metabolized in the liver and excreted primarily in the feces. | ||
==Related pages== | ==Related pages== | ||
| Line 25: | Line 25: | ||
* [[Sofosbuvir]] | * [[Sofosbuvir]] | ||
* [[Direct-acting antiviral]] | * [[Direct-acting antiviral]] | ||
[[Category:Antiviral drugs]] | [[Category:Antiviral drugs]] | ||
[[Category:Hepatitis C | [[Category:Hepatitis C treatments]] | ||
Revision as of 03:33, 13 February 2025
An antiviral medication used in the treatment of hepatitis C
| Velpatasvir | |
|---|---|
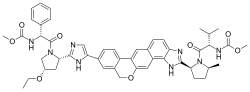
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Velpatasvir is an antiviral drug used in combination with other medications for the treatment of hepatitis C virus (HCV) infection. It is a direct-acting antiviral agent that inhibits the NS5A protein, which is essential for viral replication.
Mechanism of Action
Velpatasvir works by targeting the NS5A protein of the hepatitis C virus. NS5A is a non-structural protein that plays a crucial role in the replication of the virus and the assembly of the viral particle. By inhibiting NS5A, velpatasvir disrupts the replication cycle of HCV, thereby reducing the viral load in the body.
Clinical Use
Velpatasvir is commonly used in combination with sofosbuvir, another antiviral medication, to form a fixed-dose combination known as sofosbuvir/velpatasvir. This combination is effective against all six major genotypes of hepatitis C, making it a pan-genotypic treatment option. It is typically administered as a once-daily oral tablet.
Side Effects
Common side effects of velpatasvir, especially when used in combination with sofosbuvir, include headache, fatigue, and nausea. Serious side effects are rare but may include bradycardia when used with amiodarone.
Pharmacokinetics
Velpatasvir is well absorbed when taken orally, with peak plasma concentrations occurring approximately 3 hours after dosing. It is metabolized in the liver and excreted primarily in the feces.