Velpatasvir: Difference between revisions
No edit summary |
CSV import |
||
| Line 1: | Line 1: | ||
{{ | {{Short description|Antiviral medication used to treat hepatitis C}} | ||
{{Drugbox | |||
| verifiedfields = changed | |||
| verifiedrevid = 477002123 | |||
| image = Velpatasvir.svg | |||
| width = 200px | |||
}} | |||
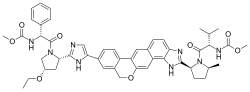
'''Velpatasvir''' is an [[antiviral drug]] used in combination with [[sofosbuvir]] to treat [[hepatitis C]] virus (HCV) infection. It is a [[direct-acting antiviral]] agent that works by inhibiting the [[NS5A protein]], which is essential for viral replication. | |||
==Medical uses== | |||
Velpatasvir is used in combination with sofosbuvir as a fixed-dose combination tablet for the treatment of chronic hepatitis C virus infection in adults. This combination is effective against all six major genotypes of HCV, making it a [[pan-genotypic]] treatment option. The standard treatment duration is 12 weeks, but it may vary depending on the presence of [[cirrhosis]] or previous treatment history. | |||
==Mechanism of action== | |||
Velpatasvir is an inhibitor of the NS5A protein, a non-structural protein of the hepatitis C virus. NS5A is involved in viral replication and assembly, and its inhibition disrupts these processes, leading to a reduction in viral load. When used with sofosbuvir, which inhibits the NS5B polymerase, the combination effectively halts the replication of the virus. | |||
==Side effects== | |||
Common side effects of the velpatasvir and sofosbuvir combination include headache, fatigue, and nausea. Serious side effects are rare but may include bradycardia when used with [[amiodarone]]. Patients are advised to consult their healthcare provider if they experience any unusual symptoms. | |||
==Pharmacokinetics== | |||
Velpatasvir is well absorbed orally, with peak plasma concentrations occurring approximately 3 hours after dosing. It is highly protein-bound and is metabolized in the liver. The drug is primarily excreted in the feces, with a small amount eliminated in the urine. | |||
==History== | |||
Velpatasvir was developed by [[Gilead Sciences]] and was approved by the [[U.S. Food and Drug Administration]] (FDA) in 2016 as part of the fixed-dose combination with sofosbuvir, marketed under the brand name [[Epclusa]]. It was the first pan-genotypic treatment approved for hepatitis C. | |||
==Research== | |||
Ongoing research is exploring the use of velpatasvir in combination with other antiviral agents to improve treatment outcomes and reduce treatment duration. Studies are also investigating its efficacy in special populations, such as those with [[HIV]] co-infection or [[renal impairment]]. | |||
==Related pages== | |||
* [[Hepatitis C]] | |||
* [[Sofosbuvir]] | |||
* [[Direct-acting antiviral]] | |||
{{ | ==References== | ||
{{Reflist}} | |||
[[Category:Antiviral drugs]] | |||
[[Category:Hepatitis C]] | |||
[[Category:Gilead Sciences]] | |||
Revision as of 11:59, 9 February 2025
Antiviral medication used to treat hepatitis C
| Velpatasvir | |
|---|---|
| |
| INN | |
| Drug class | |
| Routes of administration | |
| Pregnancy category | |
| Bioavailability | |
| Metabolism | |
| Elimination half-life | |
| Excretion | |
| Legal status | |
| CAS Number | |
| PubChem | |
| DrugBank | |
| ChemSpider | |
| KEGG | |
Velpatasvir is an antiviral drug used in combination with sofosbuvir to treat hepatitis C virus (HCV) infection. It is a direct-acting antiviral agent that works by inhibiting the NS5A protein, which is essential for viral replication.
Medical uses
Velpatasvir is used in combination with sofosbuvir as a fixed-dose combination tablet for the treatment of chronic hepatitis C virus infection in adults. This combination is effective against all six major genotypes of HCV, making it a pan-genotypic treatment option. The standard treatment duration is 12 weeks, but it may vary depending on the presence of cirrhosis or previous treatment history.
Mechanism of action
Velpatasvir is an inhibitor of the NS5A protein, a non-structural protein of the hepatitis C virus. NS5A is involved in viral replication and assembly, and its inhibition disrupts these processes, leading to a reduction in viral load. When used with sofosbuvir, which inhibits the NS5B polymerase, the combination effectively halts the replication of the virus.
Side effects
Common side effects of the velpatasvir and sofosbuvir combination include headache, fatigue, and nausea. Serious side effects are rare but may include bradycardia when used with amiodarone. Patients are advised to consult their healthcare provider if they experience any unusual symptoms.
Pharmacokinetics
Velpatasvir is well absorbed orally, with peak plasma concentrations occurring approximately 3 hours after dosing. It is highly protein-bound and is metabolized in the liver. The drug is primarily excreted in the feces, with a small amount eliminated in the urine.
History
Velpatasvir was developed by Gilead Sciences and was approved by the U.S. Food and Drug Administration (FDA) in 2016 as part of the fixed-dose combination with sofosbuvir, marketed under the brand name Epclusa. It was the first pan-genotypic treatment approved for hepatitis C.
Research
Ongoing research is exploring the use of velpatasvir in combination with other antiviral agents to improve treatment outcomes and reduce treatment duration. Studies are also investigating its efficacy in special populations, such as those with HIV co-infection or renal impairment.
Related pages
References
<references group="" responsive="1"></references>