Bohemic acid: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 23: | Line 23: | ||
{{pharmacology-stub}} | {{pharmacology-stub}} | ||
==Bohemic_acid== | |||
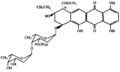
<gallery> | |||
File:Alcindoromycin.svg|Alcindoromycin structure | |||
File:Bohemanine.PNG|Bohemanine structure | |||
File:Musettamycin.PNG|Musettamycin structure | |||
File:Marcellomycin.PNG|Marcellomycin structure | |||
File:Rudolphomycin.PNG|Rudolphomycin structure | |||
File:Schaunardimycin.PNG|Schaunardimycin structure | |||
</gallery> | |||
Latest revision as of 11:15, 18 February 2025
Bohemic acid is a chemical compound that belongs to the class of organic compounds known as phenols. It is a naturally occurring substance found in certain types of fungi, including species of the genus Bohemia.
Chemical Structure[edit]
Bohemic acid is a monocyclic compound with a single benzene ring in its structure. It has a molecular formula of C7H6O3 and a molecular weight of 138.12 g/mol. The compound is characterized by the presence of a hydroxyl group (-OH) attached to the benzene ring, which is responsible for its phenolic properties.
Properties[edit]
Bohemic acid is a white, crystalline solid at room temperature. It is soluble in water and most organic solvents, including ethanol, methanol, and acetone. The compound has a melting point of 132-134 °C and a boiling point of 310 °C. It is stable under normal conditions but can decompose upon exposure to light or heat, producing toxic fumes of carbon monoxide and carbon dioxide.
Biological Activity[edit]
Bohemic acid has been studied for its potential antimicrobial and antifungal activities. Some studies have shown that it can inhibit the growth of certain types of bacteria and fungi, including Staphylococcus aureus and Candida albicans. However, further research is needed to confirm these findings and to determine the exact mechanisms of action of bohemic acid.
Safety[edit]
As with all phenolic compounds, bohemic acid should be handled with care. It can cause irritation to the skin and eyes, and prolonged exposure can lead to serious health effects. Always use appropriate personal protective equipment when handling bohemic acid, and follow all safety guidelines and regulations.
See Also[edit]
Bohemic_acid[edit]
-
Alcindoromycin structure
-
Bohemanine structure
-
Musettamycin structure
-
Marcellomycin structure
-
Rudolphomycin structure
-
Schaunardimycin structure