Cyclooctatetraene: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 31: | Line 31: | ||
{{chemistry-stub}} | {{chemistry-stub}} | ||
{{medicine-stub}} | {{medicine-stub}} | ||
<gallery> | |||
File:Willstaetter Synthesis COT.svg|Willstaetter Synthesis COT | |||
File:All-Z-Cyclooctatetraene 3D skeletal formula.svg|All-Z Cyclooctatetraene 3D skeletal formula | |||
File:Reppe Synthesis COT.svg|Reppe Synthesis COT | |||
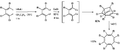
File:NewSemibullvaleneSynthesis.png|New Semibullvalene Synthesis | |||
File:Cyclooctatetraenide-3D-ball.png|Cyclooctatetraenide 3D ball | |||
File:Uranocene-3D-vdW.png|Uranocene 3D vdW | |||
File:CyclobutadieneirontricarbonylSynthesis.png|Cyclobutadieneirontricarbonyl Synthesis | |||
</gallery> | |||
Latest revision as of 05:12, 3 March 2025
Cyclooctatetraene (COT) is a cyclic compound with the formula C8H8. This hydrocarbon features a ring of eight carbon atoms, with alternating single and double bonds. It is a colorless liquid that is stable at room temperature.
Structure and bonding[edit]
Cyclooctatetraene is notable for its unusual molecular geometry and electronic structure. The molecule adopts a non-planar (tub-shaped or boat-like) conformation to allow for the maximum separation of the hydrogen atoms. This is due to the fact that a planar structure would have 4n (n is an integer) π-electrons, which, according to Hückel's rule, would make it antiaromatic and highly unstable.
Synthesis[edit]
Cyclooctatetraene was first synthesized in 1911 by Richard Willstätter at the University of Munich. The most common method of synthesis today involves the dimerization of butadiene, a process known as the Diels-Alder reaction.
Reactions[edit]
Cyclooctatetraene undergoes reactions typical of an alkene. It can be hydrogenated to form cyclooctane and it can undergo addition reactions with halogens. It also reacts with transition metals to form metallocenes, which are used in catalysis and polymerization reactions.
Applications[edit]
Cyclooctatetraene and its derivatives have been used in the synthesis of organic compounds, including pharmaceuticals and polymers. It is also used as a ligand in organometallic chemistry.
Safety[edit]
Like many hydrocarbons, cyclooctatetraene is flammable. It should be stored in a cool, well-ventilated area away from sources of ignition.
See also[edit]
References[edit]
<references />
-
Willstaetter Synthesis COT
-
All-Z Cyclooctatetraene 3D skeletal formula
-
Reppe Synthesis COT
-
New Semibullvalene Synthesis
-
Cyclooctatetraenide 3D ball
-
Uranocene 3D vdW
-
Cyclobutadieneirontricarbonyl Synthesis