Isoquinoline: Difference between revisions
CSV import Tags: mobile edit mobile web edit |
CSV import |
||
| Line 29: | Line 29: | ||
{{chemistry-stub}} | {{chemistry-stub}} | ||
{{medicine-stub}} | {{medicine-stub}} | ||
<gallery> | |||
File:Isoquinoline_numbered.svg|Isoquinoline structure | |||
File:Pomeranz-Fritsch_einfach.svg|Pomeranz-Fritsch reaction | |||
File:Bischler-Naperialski_reaction_to_papaverine.svg|Bischler-Napieralski reaction to papaverine | |||
File:Pictet-Gams_reaction.svg|Pictet-Gams reaction | |||
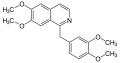
File:(S)-norcoclaurine_synthesis.svg|(S)-Norcoclaurine synthesis | |||
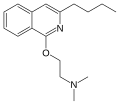
File:Quinisocaine.svg|Quinisocaine structure | |||
File:Saquinavir_structure.svg|Saquinavir structure | |||
File:Papaverin_-_Papaverine.svg|Papaverine structure | |||
File:Isoquincl.jpg|Isoquinoline | |||
</gallery> | |||
Latest revision as of 11:22, 18 February 2025
Isoquinoline is an organic compound with the formula C9H7N. It is a heterocyclic aromatic organic compound similar to benzene and pyridine, and is often used as a building block for more complex chemical compounds, including many found in natural products and pharmaceuticals.
Structure and reactivity[edit]
Isoquinoline is a colorless hygroscopic liquid at room temperature with a penetrating, unpleasant odor. The structure of isoquinoline is virtually identical to that of quinoline, but the nitrogen atom is in position 2 of the ring, not position 1. Isoquinoline is a weak base, with a pKa of 5.14. It forms salts with acids, which are usually solid and water-soluble.
Synthesis[edit]
Isoquinoline can be synthesized in a number of ways, but the most common method is the Pomeranz-Fritsch reaction, which involves the condensation of benzaldehyde with a nitrogen-containing compound.
Applications[edit]
Isoquinoline is used in the manufacture of a number of pharmaceuticals, including papaverine and laudanosine. It is also used in the production of dyes and plastics, and as a corrosion inhibitor for metals.
Health effects[edit]
Exposure to isoquinoline can cause eye and skin irritation, and prolonged exposure can lead to central nervous system depression. It is also a potential occupational carcinogen.
See also[edit]
References[edit]
<references />
-
Isoquinoline structure
-
Pomeranz-Fritsch reaction
-
Bischler-Napieralski reaction to papaverine
-
Pictet-Gams reaction
-
(S)-Norcoclaurine synthesis
-
Quinisocaine structure
-
Saquinavir structure
-
Papaverine structure
-
Isoquinoline