Zinc compounds
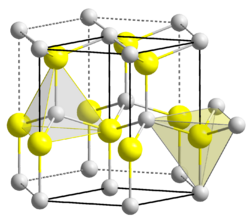
Zinc compounds are chemical compounds containing zinc, an element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a silvery-greyish appearance when oxidation is removed. It is the 24th most abundant element in the Earth's crust and has five stable isotopes. Zinc compounds are used in a wide range of applications, from medicine and pharmaceuticals to agriculture, food processing, and manufacturing.
Types of Zinc Compounds[edit]
Zinc compounds can be categorized based on their chemistry and applications. Some of the most common types include:
Zinc Oxide[edit]
Zinc oxide (ZnO) is perhaps the most widely used zinc compound, known for its role in sunscreens due to its ability to block UV light. It is also used in paints, plastics, and ceramics, and as a dietary supplement.
Zinc Sulfide[edit]
Zinc sulfide (ZnS) is used in making luminous paints, fluorescent lights, and x-ray screens. It is also a common component in the manufacturing of pigments and rubber.
Zinc Chloride[edit]
Zinc chloride (ZnCl2) is employed in wood preservation, deodorants, and embalming fluids. It is also used as a catalyst in the synthesis of various chemical compounds.
Zinc Sulfate[edit]
Zinc sulfate (ZnSO4) is used as a dietary supplement to prevent zinc deficiency and to treat acne, due to its anti-inflammatory properties. It is also used in the production of rayon, as a preservative for leathers and woods, and in agriculture as a micronutrient in fertilizers.
Health and Environmental Impact[edit]
Zinc is an essential trace element for humans, animals, and plants. It plays a crucial role in various biological processes, including enzyme function, protein synthesis, and cellular metabolism. However, excessive exposure to zinc compounds can lead to zinc toxicity, which can cause nausea, vomiting, loss of appetite, stomach cramps, diarrhea, and headaches. Environmental exposure to high levels of zinc, particularly in aquatic environments, can be harmful to fish and other wildlife.
Applications[edit]
Zinc compounds have a wide range of applications across various industries:
- In medicine, zinc oxide is used in ointments for treating minor skin conditions and as a natural sunscreen. Zinc sulfate is used as a supplement to treat zinc deficiency.
- In agriculture, zinc compounds are used as micronutrient fertilizers to correct zinc deficiency in crops.
- In manufacturing, zinc compounds are used in the production of tires, rubber products, paints, and ceramics.
- In food processing, zinc compounds are used as food additives and in packaging materials to prevent spoilage.
Conclusion[edit]
Zinc compounds play a vital role in various sectors due to their diverse properties and applications. While they are essential for health and have numerous industrial uses, it is important to manage and monitor their use to prevent environmental damage and health risks associated with overexposure.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
