Water (data page)




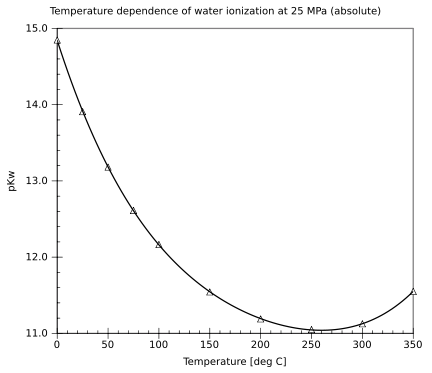 |Temperature dependence water ionization.svg]]|thumb]] Water (data page)
|Temperature dependence water ionization.svg]]|thumb]] Water (data page)
Water, a chemical compound with the formula H2O, is a transparent, tasteless, odorless, and nearly colorless chemical substance, which is the main constituent of Earth's hydrosphere and the fluids of all known living organisms. It is vital for all known forms of life, even though it provides no calories or organic nutrients. This data page focuses on the physical and chemical properties of water, along with its role in the environment and technology.
Properties[edit]
Water has unique properties that make it an essential compound for life. It is a polar molecule, with the oxygen atom having a slight negative charge and the hydrogen atoms a slight positive charge. This polarity allows it to form hydrogen bonds, which are responsible for many of its special properties, including its high surface tension, high boiling point, and ability to dissolve many substances.
Physical Properties[edit]
- Boiling Point: 100°C (212°F) at 1 atmosphere pressure
- Freezing Point: 0°C (32°F) at 1 atmosphere pressure
- Density: 1 g/cm³ at 4°C (39°F), density decreases when frozen
- pH: Pure water has a pH of 7, making it neutral
Chemical Properties[edit]
- Molecular Weight: 18.01528 g/mol
- Solvent Properties: Water is known as the "universal solvent" because it dissolves more substances than any other liquid. This property is crucial for the transport of nutrients and waste in biological organisms.
Distribution in Nature[edit]
Water covers about 71% of the Earth's surface, mainly in seas and oceans. Fresh water is found in rivers, lakes, and underground aquifers, but it constitutes only a small fraction of the Earth's total water supply.
Role in the Environment[edit]
Water plays a key role in the environment by regulating the climate through its large heat capacity and by transporting heat through the atmosphere and oceans. It is also essential for the water cycle, which includes processes such as evaporation, condensation, precipitation, and runoff.
Uses[edit]
Water is used in various ways, including as a drinking water, in agriculture for irrigation, in industry as a coolant or solvent, and in energy production in hydroelectric power plants.
Water Quality and Treatment[edit]
Water quality is crucial for health, agriculture, and industry. Treatment processes such as filtration, sedimentation, and disinfection are used to remove contaminants and make water safe for consumption and use.
Challenges[edit]
Access to clean water remains a challenge in many parts of the world, leading to water scarcity and pollution problems. Efforts to improve water management, conservation, and treatment are ongoing to ensure sustainable water resources for future generations.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
