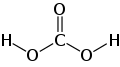
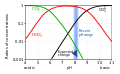
Carbonic acid
Carbonic Acid is a type of acid that is formed when carbon dioxide (CO2) is dissolved in water (H2O). It is a weak acid and is a key component in the carbon cycle, playing a crucial role in photosynthesis and respiration in plants and animals.
Chemical Composition[edit]
Carbonic acid is composed of one carbon atom, two oxygen atoms, and one hydrogen atom, giving it the chemical formula H2CO3. It is formed when carbon dioxide reacts with water, a process that can be represented by the following chemical equation:
CO2 + H2O ↔ H2CO3
Properties[edit]
Carbonic acid is a weak acid, meaning it does not fully dissociate in water. It is also a diprotic acid, meaning it can donate two protons per molecule. The first dissociation produces a bicarbonate ion (HCO3-) and a hydronium ion (H3O+), while the second dissociation produces a carbonate ion (CO3 2-) and another hydronium ion.
Role in Nature[edit]
Carbonic acid plays a vital role in the carbon cycle. It is produced in the bodies of animals during respiration, where it is exhaled as carbon dioxide. In the ocean, it helps to regulate the pH of seawater and is a key component in the process of ocean acidification. In plants, it is used in the process of photosynthesis to produce glucose and oxygen.
Health and Safety[edit]
While carbonic acid is generally safe, it can be harmful in large amounts. It is a mild irritant to the skin and eyes, and can cause respiratory problems if inhaled in high concentrations. It is also a key contributor to the acidification of the ocean, which can have detrimental effects on marine life.
See Also[edit]
-
Structural formula of carbonic acid
-
Speciation of a weak acid
-
Carbonate system of seawater
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian