Troriluzole
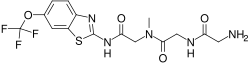
Troriluzole is a novel investigational drug that is being studied for its potential therapeutic effects in various neurological and psychiatric disorders. It is a prodrug of riluzole, which is an established medication used primarily in the treatment of amyotrophic lateral sclerosis (ALS).
Mechanism of Action[edit]
Troriluzole is designed to modulate the glutamatergic system by reducing the synaptic release of glutamate, an excitatory neurotransmitter. Excessive glutamate activity is implicated in several neurological conditions, and by modulating this pathway, troriluzole aims to provide neuroprotective effects.
Clinical Applications[edit]
Troriluzole is currently being investigated for its efficacy in treating a variety of conditions, including:
- Generalized anxiety disorder (GAD)
- Obsessive-compulsive disorder (OCD)
- Spinocerebellar ataxia (SCA)
- Alzheimer's disease
Generalized Anxiety Disorder[edit]
In clinical trials, troriluzole has shown promise in reducing symptoms of generalized anxiety disorder by modulating glutamate levels, which are often dysregulated in anxiety disorders.
Obsessive-Compulsive Disorder[edit]
Troriluzole is also being studied for its potential to alleviate symptoms of obsessive-compulsive disorder. The drug's ability to modulate glutamate may help in reducing the compulsive behaviors and obsessive thoughts characteristic of OCD.
Spinocerebellar Ataxia[edit]
Spinocerebellar ataxia is a group of genetic disorders characterized by progressive problems with movement. Troriluzole is being evaluated for its potential to slow the progression of these disorders by protecting neurons from glutamate-induced excitotoxicity.
Alzheimer's Disease[edit]
Alzheimer's disease is a neurodegenerative disorder marked by cognitive decline. Troriluzole is under investigation for its potential to improve cognitive function and slow disease progression by modulating glutamate activity.
Development and Trials[edit]
Troriluzole is being developed by Biohaven Pharmaceuticals. The drug has undergone several phases of clinical trials to evaluate its safety, tolerability, and efficacy in various conditions. Results from these trials will determine its potential for approval and use in clinical practice.
See Also[edit]
References[edit]
External Links[edit]
-
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian