Trimethyltrienolone
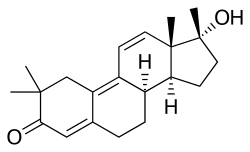
Trimethyltrienolone, also known by its developmental code name R-2956, is a synthetic, orally active androgen and anabolic steroid (AAS) and a derivative of nandrolone (19-nortestosterone). It is specifically a derivative of trenbolone (trienolone), a potent AAS, with the addition of three methyl groups at the R2, R17, and R18 positions. This modification significantly increases its oral bioavailability and androgenic potency, making it one of the most potent AAS known.
Pharmacology[edit]
Trimethyltrienolone binds strongly to the androgen receptor (AR), much more so than testosterone and nandrolone. Its affinity for the AR is said to be one of the highest among synthetic AAS. This high affinity is attributed to the structural modifications it has undergone, particularly the addition of methyl groups, which protect the molecule from metabolic breakdown in the liver, allowing a significant portion to enter the bloodstream intact. Its potent androgenic effects are accompanied by strong anabolic effects, though the exact ratio of androgenic to anabolic activity is not well-defined but is believed to be quite high.
Clinical Use[edit]
Despite its potent properties, trimethyltrienolone has never been marketed for medical use. Its development was likely halted due to the potential for severe hepatotoxicity (liver toxicity) and other side effects associated with its strong androgenic activity and oral bioavailability. As such, its use has been limited to scientific research, including studies on the androgen receptor and the effects of androgens on muscle and bone tissues.
Side Effects[edit]
Given its potent androgenic properties, trimethyltrienolone can cause severe side effects, particularly those related to its androgenic effects. These can include acne, hair loss, virilization in women (development of male characteristics), and an increased risk of prostate cancer in men. Additionally, its structural modifications that enhance oral bioavailability also make it potentially hepatotoxic, posing a significant risk to liver health.
Bodybuilding and Athletic Use[edit]
Like many other AAS, trimethyltrienolone has found a place in the bodybuilding and athletic communities, despite its lack of medical approval. It is prized for its potent anabolic effects, which can significantly increase muscle mass and strength. However, due to its strong androgenic effects and potential for severe liver toxicity, it is considered a high-risk substance and is not recommended for use. Its possession, sale, and use are also illegal in many countries under laws regulating controlled substances.
Legal Status[edit]
Trimethyltrienolone is classified as a controlled substance in many countries, making its non-prescribed possession, sale, or use illegal. The legal status varies by country, but it is generally included in the same categories as other potent AAS, reflecting concerns over its potential for abuse, health risks, and lack of approved medical use.
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian