Adenosine triphosphate
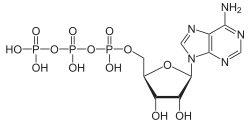
Adenosine triphosphate (ATP) is a complex organic chemical that provides energy to drive many processes in living cells, such as muscle contraction, nerve impulse propagation, and chemical synthesis. Found in all forms of life, ATP is often referred to as the "molecular unit of currency" of intracellular energy transfer.
Structure[edit]

ATP consists of an adenosine molecule bonded to three phosphate groups. The adenosine molecule is composed of an adenine ring and a ribose sugar. The three phosphate groups are labeled alpha, beta, and gamma, starting with the group closest to the ribose. The bonds between these phosphate groups are high-energy bonds, which release energy when broken.
Function[edit]
ATP is used by cells as a coenzyme in various biochemical reactions. It is involved in metabolism, serving as a substrate for enzymes and signal transduction pathways. ATP is also crucial in DNA and RNA synthesis, where it acts as a building block.
Energy Transfer[edit]

The energy stored in ATP is released when it is hydrolyzed to adenosine diphosphate (ADP) and an inorganic phosphate. This reaction releases energy that can be used by the cell to perform work. The conversion of ATP to ADP is a reversible process, allowing ATP to be regenerated from ADP and phosphate through cellular respiration.
Role in Cellular Processes[edit]
ATP is essential for many cellular processes, including:
- Muscle contraction: ATP binds to myosin, allowing it to interact with actin filaments and produce muscle contraction.
- Active transport: ATP provides energy for the active transport of molecules across cell membranes.
- Signal transduction: ATP is involved in signaling pathways, acting as a substrate for kinases that phosphorylate proteins.
ATP in Metabolism[edit]
ATP is produced through various metabolic pathways, including:
- Glycolysis: A process that breaks down glucose to produce ATP.
- Citric acid cycle: Also known as the Krebs cycle, it generates ATP through the oxidation of acetyl-CoA.
- Oxidative phosphorylation: Occurs in the mitochondria, where ATP is produced from the electron transport chain.
ATP Binding and Protein Interaction[edit]

ATP binds to various proteins, often at specific motifs such as the Rossmann fold, which is a common structural motif in proteins that bind nucleotides like ATP.
Magnesium and ATP[edit]

Magnesium ions (Mg²⁺) are essential for the function of ATP. They stabilize the negative charges on the phosphate groups, facilitating the binding of ATP to enzymes and other proteins.
Related Pages[edit]
Ad. Transform your life with W8MD's Budget GLP-1 injections from $29.99


W8MD offers medical weight loss programs including NYC medical weight loss and Philadelphia medical weight loss offering:
- Affordable GLP1 shots (generic and brand names) such as
- Wegovy NYC (Semaglutide)
- Zepbound NYC /
- Learn more: Budget GLP1 weight loss injections NYC & Philadelphia GLP1 weight loss shots
- Most insurances accepted
- Lowest cost GLP1 weight loss NYC such as Semaglutide starting from $29.99/week and $45.00/week (Tirzepatide) with insurance.
- Prescription weight loss NYC including:
NYC weight loss doctor appointmentsNYC weight loss doctor appointments
Start your physician weight loss journey today at our:
- NYC medical weight loss
- Philadelphia medical weight loss
- Call 718-946-5500 for NYC or 215-676-2334 for Philadelphia
Tags: Budget glp1 weight loss NYC, Zepbound NYC, Philadelphia medical weight loss, Wegovy NYC, Affordable glp1 shots Philadelphia
![]()
![]()
![]() Google plus
Google plus
Advertise on WikiMD
|
WikiMD's Wellness Encyclopedia |
| Let Food Be Thy Medicine Medicine Thy Food - Hippocrates |
Medical Disclaimer: WikiMD is not a substitute for professional medical advice. The information on WikiMD is provided as an information resource only, may be incorrect, outdated or misleading, and is not to be used or relied on for any diagnostic or treatment purposes. Please consult your health care provider before making any healthcare decisions or for guidance about a specific medical condition. WikiMD expressly disclaims responsibility, and shall have no liability, for any damages, loss, injury, or liability whatsoever suffered as a result of your reliance on the information contained in this site. By visiting this site you agree to the foregoing terms and conditions, which may from time to time be changed or supplemented by WikiMD. If you do not agree to the foregoing terms and conditions, you should not enter or use this site. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian