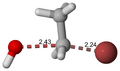
Transition state
Concept in chemistry
Transition state is a key concept in chemical kinetics and reaction mechanisms. It refers to a particular configuration along the reaction coordinate. It is the state corresponding to the highest energy along this path. The transition state is often marked by the formation of a high-energy, unstable arrangement of atoms that exists momentarily as reactants are converted into products.
Theory[edit]
The concept of the transition state is central to the transition state theory (TST), which provides a framework for understanding how chemical reactions occur and how reaction rates are determined. According to TST, the transition state is a transient configuration of atoms at the peak of the potential energy barrier separating reactants and products. The energy required to reach this state is known as the activation energy.
Characteristics[edit]
The transition state is characterized by a partial bond formation and bond breaking. It is not a stable state and cannot be isolated. Instead, it is a fleeting moment in the reaction pathway. The structure of the transition state can be inferred using various experimental techniques and computational methods, such as X-ray crystallography and quantum chemistry calculations.
Examples[edit]
One classic example of a transition state is found in the [[SN2 reaction]] mechanism. In the reaction of bromoethane with a nucleophile, the transition state involves a pentavalent carbon atom where the nucleophile and the leaving group are both partially bonded to the carbon.
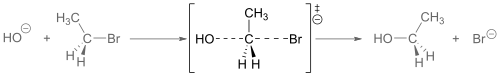
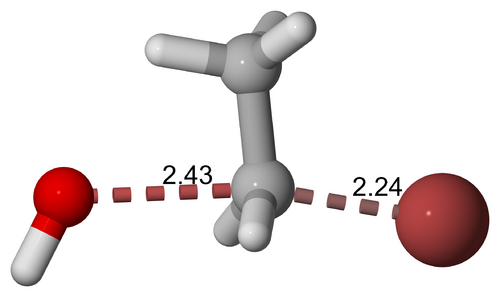
Another example is the base hydrolysis of bromoethane, where the transition state involves the simultaneous breaking of the C-Br bond and the formation of the C-OH bond.
Structure Correlation Principle[edit]
The Structure Correlation Principle is a concept that relates the structure of the transition state to the structure of the reactants and products. It suggests that the transition state resembles the structure of the species to which it is more similar in energy.

Related pages[edit]
- Reaction mechanism
- Activation energy
- Transition state theory
- [[SN2 reaction]]
References[edit]
- Laidler, K. J. (1987). "Chemical Kinetics". Harper & Row.
- Anslyn, E. V., & Dougherty, D. A. (2006). "Modern Physical Organic Chemistry". University Science Books.
-
Bromoethane SN2 reaction
-
Base hydrolysis of bromoethane, transition state
-
Structure Correlation Principle
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian