Activation energy
Energy required to start a chemical reaction
Activation energy is the minimum amount of energy that must be provided to compounds to result in a chemical reaction. It is a crucial concept in the field of chemical kinetics and plays a significant role in determining the rate at which a reaction proceeds.
Overview[edit]
Activation energy is often denoted by the symbol E_. It represents the energy barrier that must be overcome for reactants to be transformed into products. This concept is applicable to both endothermic and exothermic reactions.
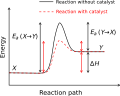
In a potential energy surface, the activation energy is the difference in energy between the reactants and the highest energy transition state. The transition state is a temporary, high-energy configuration that reactants must pass through to become products.
Arrhenius Equation[edit]
The relationship between the activation energy and the rate of a chemical reaction is described by the Arrhenius equation:
- k = A e^{-E_/RT}
where:
- k is the rate constant of the reaction,
- A is the pre-exponential factor, a constant for each chemical reaction,
- E_ is the activation energy,
- R is the universal gas constant,
- T is the temperature in Kelvin.
This equation shows that the rate of a reaction increases exponentially with a decrease in activation energy or an increase in temperature.
Catalysts[edit]
Catalysts are substances that increase the rate of a chemical reaction by lowering the activation energy. They provide an alternative reaction pathway with a lower energy barrier. Importantly, catalysts are not consumed in the reaction and do not alter the equilibrium position.
Applications[edit]
Understanding activation energy is essential in various fields such as biochemistry, pharmacology, and materials science. For instance, in biochemistry, enzymes act as biological catalysts to lower the activation energy of biochemical reactions, facilitating vital processes in living organisms.
Visual Representation[edit]
Activation energy can be visualized using energy diagrams, which plot the energy of the system as a function of the reaction coordinate. These diagrams illustrate the energy barrier that must be overcome for a reaction to proceed.
Related pages[edit]
Gallery[edit]
-
Incandescence, a process involving activation energy.
-
Diagram illustrating activation energy.
-
Energy diagram showing activation energy.
-
Incandescence
-
Activation Energy Diagram
-
Activation Energy Curve
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian