Thioacetamide
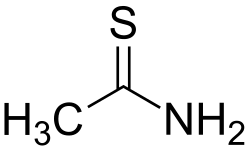
Thioacetamide is an organosulfur compound with the chemical formula C₂H₅NS. It is a white crystalline solid that is soluble in water and commonly used as a laboratory reagent and in the synthesis of other chemicals.
Chemical Properties[edit]
Thioacetamide is structurally similar to acetamide, with the oxygen atom replaced by a sulfur atom. This substitution imparts different chemical properties to thioacetamide compared to acetamide. Thioacetamide can act as a source of hydrogen sulfide (H₂S) when hydrolyzed.
Synthesis[edit]
Thioacetamide can be synthesized by the reaction of acetonitrile with hydrogen sulfide in the presence of a base. The general reaction is: CH₃CN + H₂S → CH₃C(S)NH₂
Uses[edit]
Thioacetamide is used in various applications, including:
- As a laboratory reagent for the detection of metal ions.
- In the synthesis of thioamides and other sulfur-containing compounds.
- As a source of hydrogen sulfide in chemical reactions.
Toxicity and Safety[edit]
Thioacetamide is known to be toxic and a potential carcinogen. It can cause liver damage and other health issues upon exposure. Proper safety measures, including the use of personal protective equipment (PPE) and working in a well-ventilated area, are essential when handling thioacetamide.
Environmental Impact[edit]
Thioacetamide can have detrimental effects on the environment if not disposed of properly. It is important to follow appropriate disposal guidelines to minimize its impact on the environment.
See Also[edit]
References[edit]
External Links[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
