Taxine alkaloids
Taxine alkaloids are a group of alkaloids that are found in the yew tree. They are highly toxic and have been used in the past for their poisonous properties.
Overview[edit]
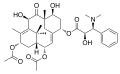
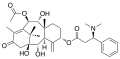
Taxine alkaloids are a group of around 40 different alkaloids that are found in the yew tree. They are named after the genus Taxus, which is the Latin name for yew. The most well-known of these alkaloids is taxine B, which is the most toxic.
Structure and Properties[edit]
Taxine alkaloids are complex organic compounds that contain a number of different functional groups. They are characterized by a core structure that consists of a cyclohexane ring, a cycloheptane ring, and a cyclooctane ring. The different taxine alkaloids are distinguished by the various substituents that are attached to this core structure.
Toxicity[edit]
Taxine alkaloids are highly toxic. They work by blocking sodium channels in the heart, which disrupts the normal electrical activity and can lead to cardiac arrest. Symptoms of taxine poisoning include dizziness, nausea, abdominal pain, and a slow or irregular heartbeat. In severe cases, it can be fatal.
Medical Use[edit]
Despite their toxicity, taxine alkaloids have been investigated for their potential use in medicine. They have been found to have anti-cancer properties, and are used in the chemotherapy drug paclitaxel (also known as Taxol). However, their use is limited by their high toxicity and the difficulty of isolating them from the yew tree.
See Also[edit]
-
Taxine A structure
-
Taxine B structure
-
Taxus baccata
-
Taxus baccata seed
-
3,5-Dimethoxyphenol structure
-
Taxine B structure
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian