Sulfuryl fluoride
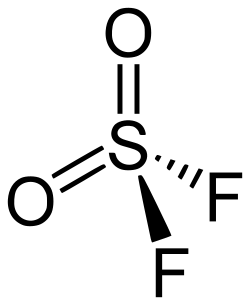
0Sulfuryl fluoride is an inorganic compound that is used primarily as a fumigant in pest control. It is a colorless and odorless gas that is non-flammable and non-corrosive.
Chemical Properties[edit]
Sulfuryl fluoride is a compound of sulfur, oxygen, and fluorine with the chemical formula SO2F2. It is a potent greenhouse gas that can contribute to global warming.
Uses[edit]
The primary use of sulfuryl fluoride is as a fumigant for the control of pests such as termites, beetles, and rats. It is also used in the food industry for the fumigation of stored grains, dried fruits, nuts, and coffee beans.
Health Effects[edit]
Exposure to sulfuryl fluoride can cause adverse health effects such as respiratory irritation, nausea, vomiting, and seizures. In severe cases, it can lead to pulmonary edema and death.
Environmental Impact[edit]
Sulfuryl fluoride is a potent greenhouse gas with a global warming potential that is significantly higher than that of carbon dioxide. It is also a significant contributor to the depletion of the ozone layer.
Regulation[edit]
In many countries, the use of sulfuryl fluoride is regulated by environmental and health agencies such as the Environmental Protection Agency in the United States and the European Chemicals Agency in the European Union.
See Also[edit]
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian
