Sodium triphosphate
Sodium Triphosphate:
```
| Sodium triphosphate | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Sodium triphosphate (STPP), also known as sodium tripolyphosphate, is a chemical compound with the formula Na5P3O10. It is a white, crystalline solid that is highly soluble in water. It is used in a wide variety of applications, including as a food preservative, a water softener, and a detergent builder.
Chemical Properties[edit]
Sodium triphosphate is a white, crystalline solid that is highly soluble in water. It has a melting point of 622 °C and a boiling point of 1,534 °C. It is a strong base and can react with acids to form salts.
Uses[edit]
Sodium triphosphate is used in a wide variety of applications. It is used as a food preservative, a water softener, and a detergent builder. It is also used in ceramics, textiles, and leather processing.
Health Effects[edit]
Exposure to sodium triphosphate can cause irritation to the eyes, skin, and respiratory tract. Ingestion can cause nausea, vomiting, and diarrhea. Long-term exposure can lead to more serious health effects, including kidney damage and osteoporosis.
See Also[edit]
References[edit]
External Links[edit]
```
Please note that this is a basic structure and you may need to add more information based on your specific needs.
-
Sodium tripolyphosphate structure
-
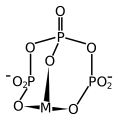
Triphosphate chelation diagram
-
Sodium triphosphate
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian