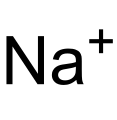Sodium chlorite
Sodium chlorite is a chemical compound used in a variety of applications, including water purification, bleaching, and manufacturing. It is a white crystalline powder that is highly soluble in water. Sodium chlorite is often used in the production of chlorine dioxide, a powerful oxidizing agent.
Chemical Properties[edit]
Sodium chlorite (NaClO2) is an inorganic salt that is produced by the reaction of chlorine dioxide with sodium hydroxide. It is a strong oxidizer and can react violently with organic materials, leading to fire or explosion. Sodium chlorite is stable under normal conditions, but it decomposes to form oxygen and sodium chloride when heated.
Uses[edit]
Sodium chlorite is used in a variety of applications due to its strong oxidizing properties. It is used in water treatment plants to kill bacteria and other microorganisms. In the paper industry, it is used as a bleaching agent. Sodium chlorite is also used in the production of disinfectants and antiseptics.
Health Effects[edit]
Exposure to sodium chlorite can cause irritation to the eyes, skin, and respiratory system. Ingestion can lead to nausea, vomiting, and abdominal pain. Long-term exposure can lead to more serious health effects, including damage to the red blood cells.
Regulation[edit]
In many countries, the use of sodium chlorite is regulated due to its potential health effects. In the United States, the Environmental Protection Agency (EPA) regulates the use of sodium chlorite in drinking water.
See Also[edit]
Sodium_chlorite[edit]
-
Sodium ion
-
Chloride ion
-
Sodium 3D model
-
Chlorite 3D van der Waals model
-
Sodium chlorite 450g
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian