Sinigrin
| Chemical Compound | |
|---|---|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider ID | |
| UNII | |
| ChEBI | |
| ChEMBL | |
| Properties | |
| Chemical Formula | |
| Molar Mass | |
| Appearance | |
| Density | |
| Melting Point | |
| Boiling Point | |
| Hazards | |
| GHS Pictograms | [[File:|50px]] |
| GHS Signal Word | |
| GHS Hazard Statements | |
| NFPA 704 | [[File:|50px]] |
| References | |
Sinigrin[edit]
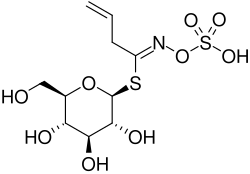
Sinigrin is a notable glucosinolate, a subclass of glucosides, primarily discovered in select plants belonging to the Brassicaceae family. Among the various sources, Brussels sprouts, broccoli, and the seeds of black mustard (Brassica nigra) are prominent carriers of this compound. The integral component of many dietary items, sinigrin is often associated with the unique pungent taste experience associated with certain foods, most notably mustard and horseradish.
Biochemical Properties[edit]
The underlying principle governing the pungency of sinigrin-rich foods revolves around its conversion to mustard oil, or allyl isothiocyanate. Whenever plants containing sinigrin sustain damage, such as when crushed or chewed, the enzyme myrosinase acts on sinigrin, facilitating its degradation into allyl isothiocyanate[1].
Seeds sourced from white mustard (Sinapis alba) yield a milder version of mustard. This is attributed to the glucosinolate sinalbin, which is distinct from sinigrin in its chemical composition and resultant taste profile.
Chemical Structure[edit]
The chemical nomenclature for sinigrin is "allylglucosinolate" or alternatively "2-propenylglucosinolate."
Significance in Nutrition and Health[edit]
The presence of sinigrin in many vegetables, especially those from the Brassicaceae family, has been subjected to numerous studies due to the potential health benefits and nutritional properties of glucosinolates. These compounds, including sinigrin, have been shown to possess antioxidant and potential anti-carcinogenic properties[2].
Conclusion[edit]
Sinigrin stands out as an intriguing compound both in terms of its dietary prevalence and potential health implications. Its characteristic pungency and associated biochemical reactions underscore the intricate relationships between food chemistry, flavor, and health.
References[edit]
- ↑ Richard, H. (Year). Arômes alimentaires. Retrieved from [1]
- ↑ Verkerk, R., Schreiner, M., Krumbein, A., Ciska, E., Holst, B., Rowland, I., ... & Dekker, M. (2009). Glucosinolates in Brassica vegetables: the influence of the food supply chain on intake, bioavailability and human health. Molecular nutrition & food research, 53(S1), S219-S265.
External links[edit]
- Institute of Food Research(link).
- Norwich Research Park website
| Glycosides | ||||||||
|---|---|---|---|---|---|---|---|---|
|
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian