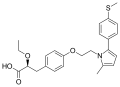
Saroglitazar
Saroglitazar is a medication primarily used for the treatment of diabetic dyslipidemia and hypertriglyceridemia. It is a dual PPAR agonist, exerting its effects on both PPAR alpha and PPAR gamma receptors. Saroglitazar was developed by Zydus Cadila, an Indian pharmaceutical company, and received approval from the Drug Controller General of India (DCGI) in June 2013.
Etymology[edit]
The name "Saroglitazar" is derived from the words "Saro" and "glitazar". "Saro" is a prefix used in pharmaceutical naming to denote a connection to the Saroglitazar family, while "glitazar" is a suffix used for drugs that act as PPAR agonists.
Pharmacology[edit]
Saroglitazar is a dual PPAR agonist, meaning it activates both the PPAR alpha and PPAR gamma receptors. These receptors play a crucial role in the regulation of lipid metabolism and glucose homeostasis, respectively. By activating these receptors, Saroglitazar helps to reduce levels of triglycerides and increase levels of HDL cholesterol, while also improving insulin sensitivity.
Clinical Use[edit]
Saroglitazar is primarily used for the treatment of diabetic dyslipidemia and hypertriglyceridemia. Diabetic dyslipidemia is a condition characterized by high levels of triglycerides and low levels of HDL cholesterol in patients with diabetes. Hypertriglyceridemia, on the other hand, is a condition characterized by excessively high levels of triglycerides in the blood.
Side Effects[edit]
Like all medications, Saroglitazar can cause side effects. The most common side effects include nausea, headache, and upper respiratory tract infection. Less common side effects can include hypoglycemia, edema, and anemia.
Related Terms[edit]
- PPAR agonist
- Diabetic dyslipidemia
- Hypertriglyceridemia
- Zydus Cadila
- Drug Controller General of India
-
Saroglitazar
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian