Ribonucleotide reductase
Ribonucleotide reductase (RNR) is an essential enzyme in all living cells, playing a pivotal role in the synthesis of deoxyribonucleotides, which are the building blocks for DNA synthesis and repair. This enzyme catalyzes the reduction of ribonucleotides, the precursors of RNA, into deoxyribonucleotides, thereby providing the necessary components for DNA replication and repair processes. Given its critical function in cell proliferation and genome maintenance, ribonucleotide reductase is a key target for anticancer drugs and antiviral agents.
Structure and Function[edit]
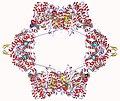
Ribonucleotide reductase is composed of two subunits, typically referred to as R1 and R2. These subunits form a complex that is required for the enzyme's catalytic activity. The R1 subunit contains the active site where the reduction of ribonucleotides takes place, while the R2 subunit contains a tyrosyl radical essential for the initiation of the catalytic process. The activity of RNR is tightly regulated through various mechanisms, including allosteric regulation and feedback inhibition, to ensure a balanced supply of deoxyribonucleotides for DNA synthesis.
Regulation[edit]
The activity of ribonucleotide reductase is regulated at multiple levels, ensuring that the production of deoxyribonucleotides is closely matched to the cell's needs. Allosteric regulation allows the enzyme to respond to the concentrations of its substrates and products, adjusting its activity accordingly. Additionally, the availability of the R2 subunit, which can be regulated through its synthesis and degradation, also plays a crucial role in controlling the overall activity of RNR.
Clinical Significance[edit]
Given its central role in DNA synthesis, ribonucleotide reductase is a prime target for therapeutic interventions aimed at controlling cell proliferation. Inhibitors of RNR, such as hydroxyurea, are used in the treatment of various cancers and conditions characterized by excessive cell proliferation. Furthermore, because of its importance in DNA replication, RNR is also a target for antiviral strategies, as viruses rely on host cell DNA synthesis machinery for their replication.
Inhibitors[edit]
Several inhibitors of ribonucleotide reductase have been identified and are used for therapeutic purposes. These inhibitors can be broadly classified into two groups: those that target the R1 subunit and those that target the R2 subunit. Inhibitors of the R1 subunit, such as hydroxyurea, act by scavenging the tyrosyl radical in the R2 subunit, thereby preventing the initiation of the catalytic process. On the other hand, inhibitors targeting the R2 subunit interfere with the formation of the essential tyrosyl radical.
Research Directions[edit]
Research on ribonucleotide reductase continues to be a vibrant field, with studies focusing on understanding the enzyme's complex regulation, discovering new inhibitors, and exploring its role in various diseases. Insights into the regulation of RNR could lead to the development of novel therapeutic strategies that selectively target cancer cells or viral infections without harming normal cells.
Ribonucleotide reductase gallery[edit]
-
3uus
-
1PEU R1E
-
PDB 1rlr EBI
-
PDB 1pem EBI
-
PDB 1rib EBI
-
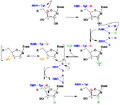
Ribonucleotide reductase mechanism
-
RNR reaction
-
Regulation of class-I ribonucleotide reductase
Medical Disclaimer: WikiMD is for informational purposes only and is not a substitute for professional medical advice. Content may be inaccurate or outdated and should not be used for diagnosis or treatment. Always consult your healthcare provider for medical decisions. Verify information with trusted sources such as CDC.gov and NIH.gov. By using this site, you agree that WikiMD is not liable for any outcomes related to its content. See full disclaimer.
Credits:Most images are courtesy of Wikimedia commons, and templates, categories Wikipedia, licensed under CC BY SA or similar.
Translate this page: - East Asian
中文,
日本,
한국어,
South Asian
हिन्दी,
தமிழ்,
తెలుగు,
Urdu,
ಕನ್ನಡ,
Southeast Asian
Indonesian,
Vietnamese,
Thai,
မြန်မာဘာသာ,
বাংলা
European
español,
Deutsch,
français,
Greek,
português do Brasil,
polski,
română,
русский,
Nederlands,
norsk,
svenska,
suomi,
Italian
Middle Eastern & African
عربى,
Turkish,
Persian,
Hebrew,
Afrikaans,
isiZulu,
Kiswahili,
Other
Bulgarian,
Hungarian,
Czech,
Swedish,
മലയാളം,
मराठी,
ਪੰਜਾਬੀ,
ગુજરાતી,
Portuguese,
Ukrainian